PUBLICATIONS
67. Angew. Chem. Int. Ed. 2026, 65, e26128
|
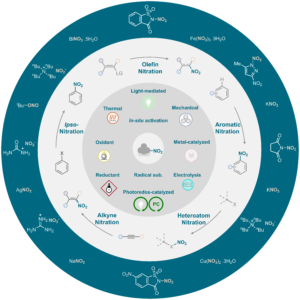
Navigating Nitration Chemistry: A Practical Guide to Reagents, Mechanisms, and Selectivity H. Lecomte, A. J. Fernandes, D. Katayev Highlighted in Synthesis Spotlight
|
66. Chem. Commun. 2026, 62, 8327-8339
|
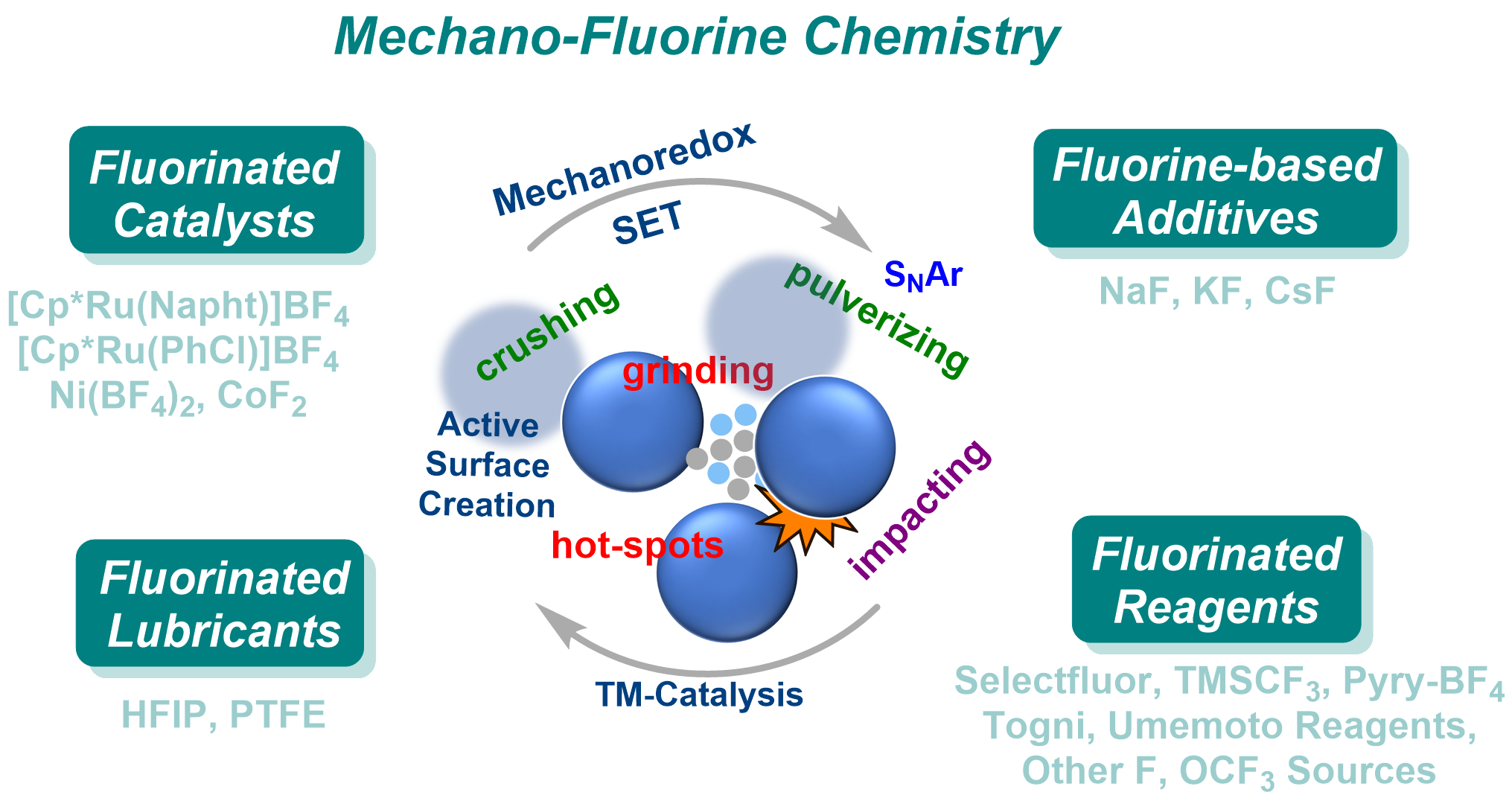
When Mechanochemistry Meets Fluorine V. B. Purohit, R. V. Prajapati, V. D. Prajapati, D. Katayev, S. Mkrtchyan, V. O. Iaroshenko
|
65. J. CO2 Util. 2026, 106, 103384
|
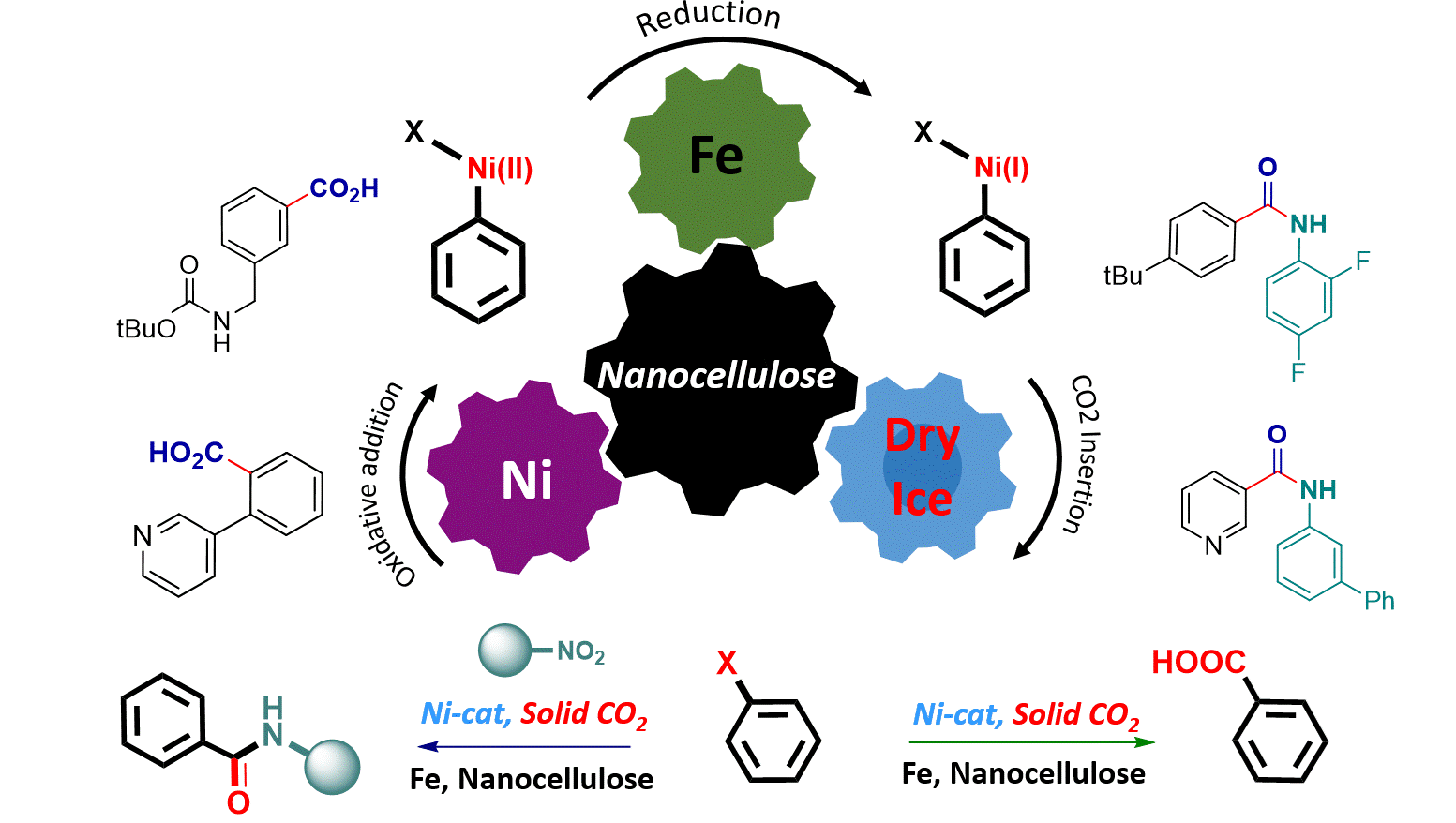
Mechanochemical synthesis of carboxylic acids and amides: Nickel-catalyzed direct carbonylation of aryl halides using dry ice S. Mkrtchyan, J. Zapletal, D. Elumalai, V. B. Purohit, R. V. Prajapati, V. D. Prajapati, G. Addová, J. Filo, B. Bielec, O. Shalimov, M. Sillanpää, D. Katayev, V. O. Iaroshenko
|
64. Org. Lett. 2026, 28, 11, 3388–3393
|
Triplet Energy Transfer-Mediated Intermolecular Paternò-Büchi Reaction for the Synthesis of Trifluoromethylated Oxetanes Y. Zhu, A. J. Fernandes, E. Zhilin, D. Katayev |

63. J. Org. Chem. 2026, 91, 8, 3138–3149
|
Electrophilic and Radical Ability of Organic Nitrating Reagents A. J. Fernandes, H. Lecomte, D. Katayev |
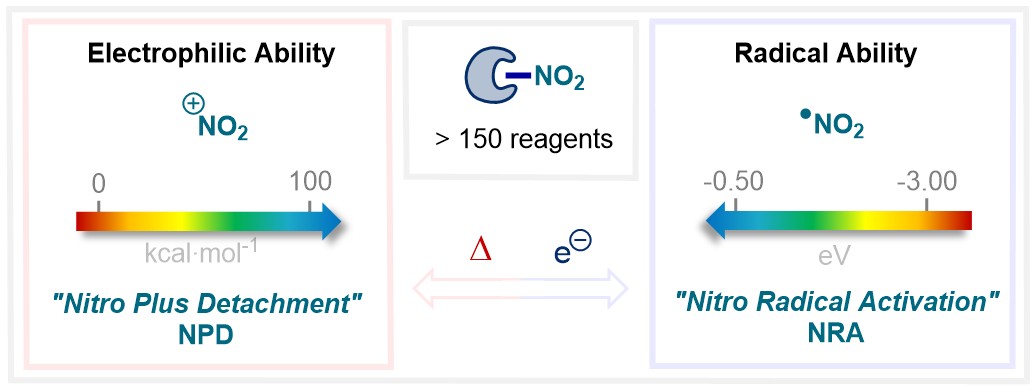
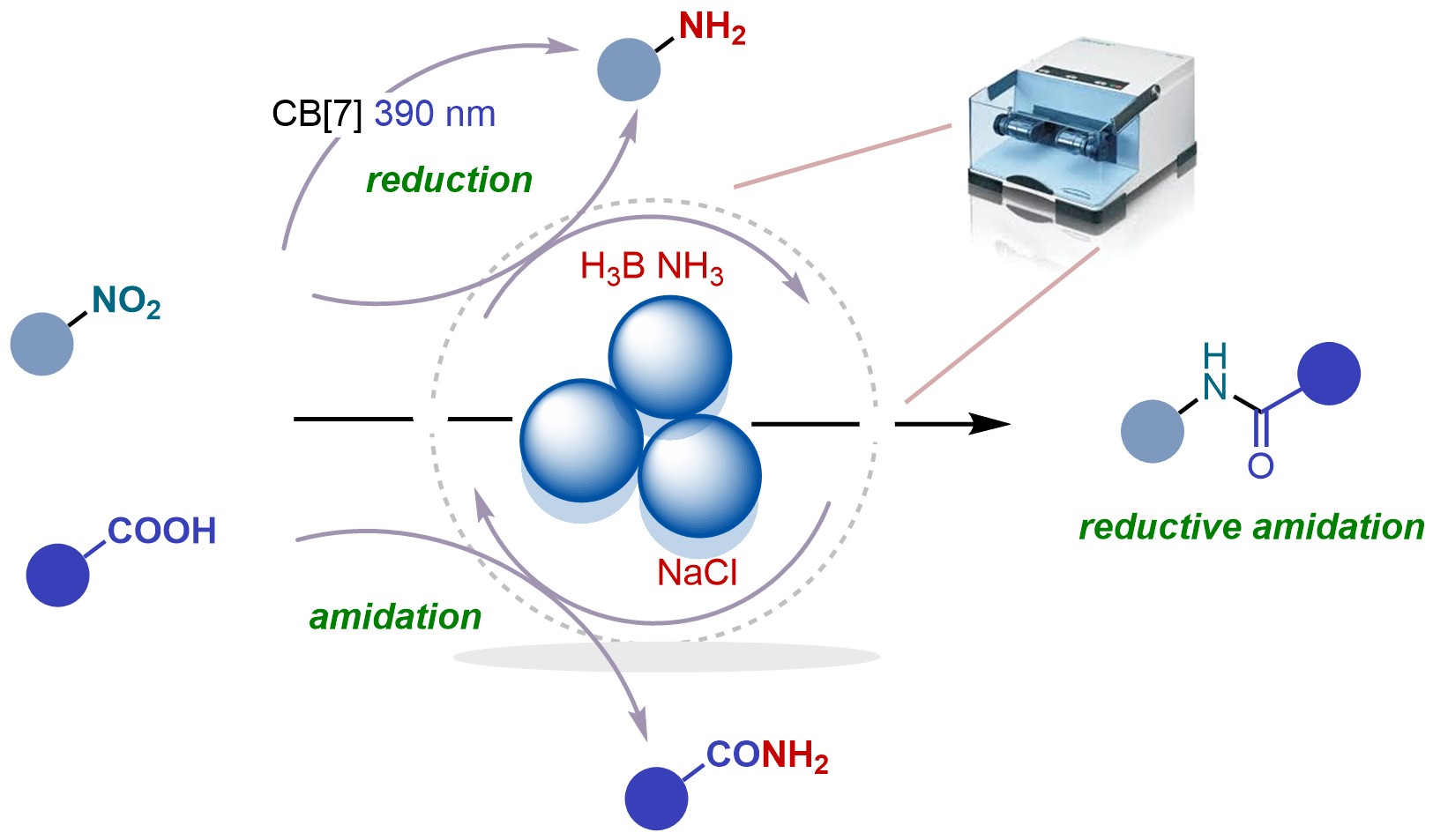
62. Org. Chem. Front. 2026, 13, 874-883
|
Photo and Mechanochemically Induced Reduction of Nitroarenes Using Ammonia Borane S. Mkrtchyan, V. B. Purohit, O. Shalimov, Y. Karpun, R. Prajapati, V. D. Prajapati, B. Bielec, G. Addova, J. Filo, P. Jin, C. Wang, D. Katayev, V. Iarochenko |
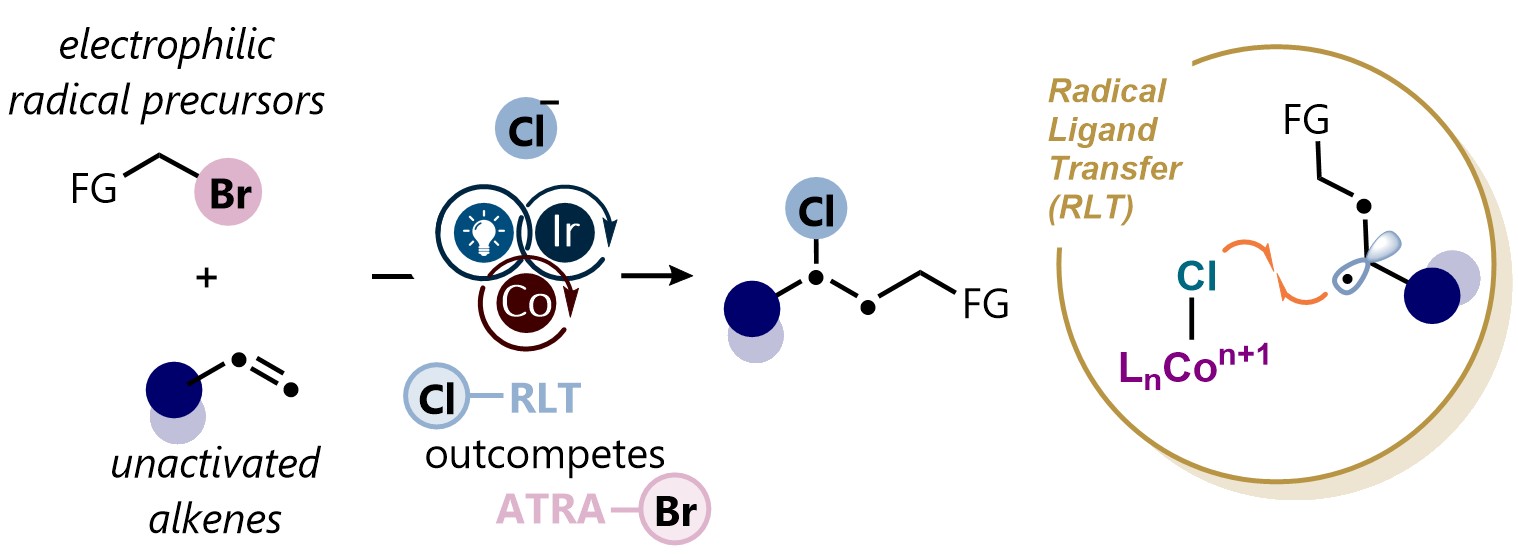
61. Jacs Au 2025, 5, 12, 6231-6240
|
Chloroalkylation of Unactivated Alkenes via Cobalt-Mediated Radical Ligand Transfer (RLT) Photoredox Catalysis Platform S. Patra, A. J. Fernandes, B. Kadriu, D. Katayev |
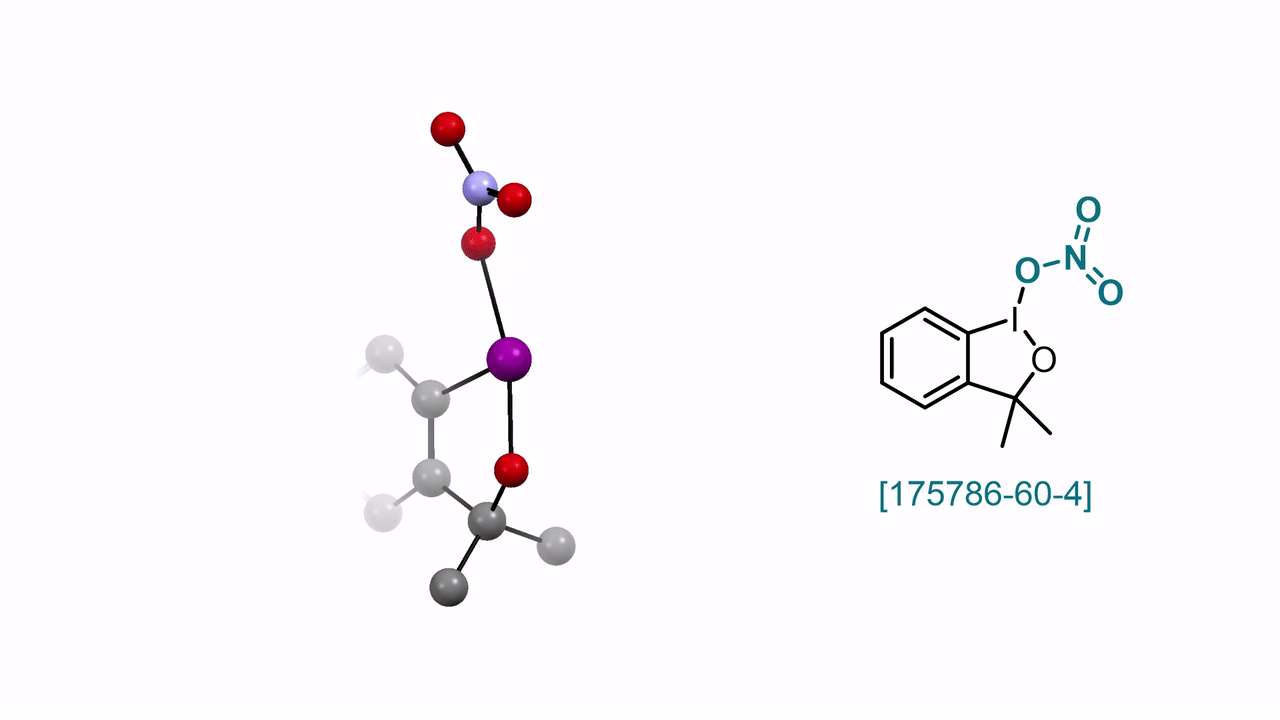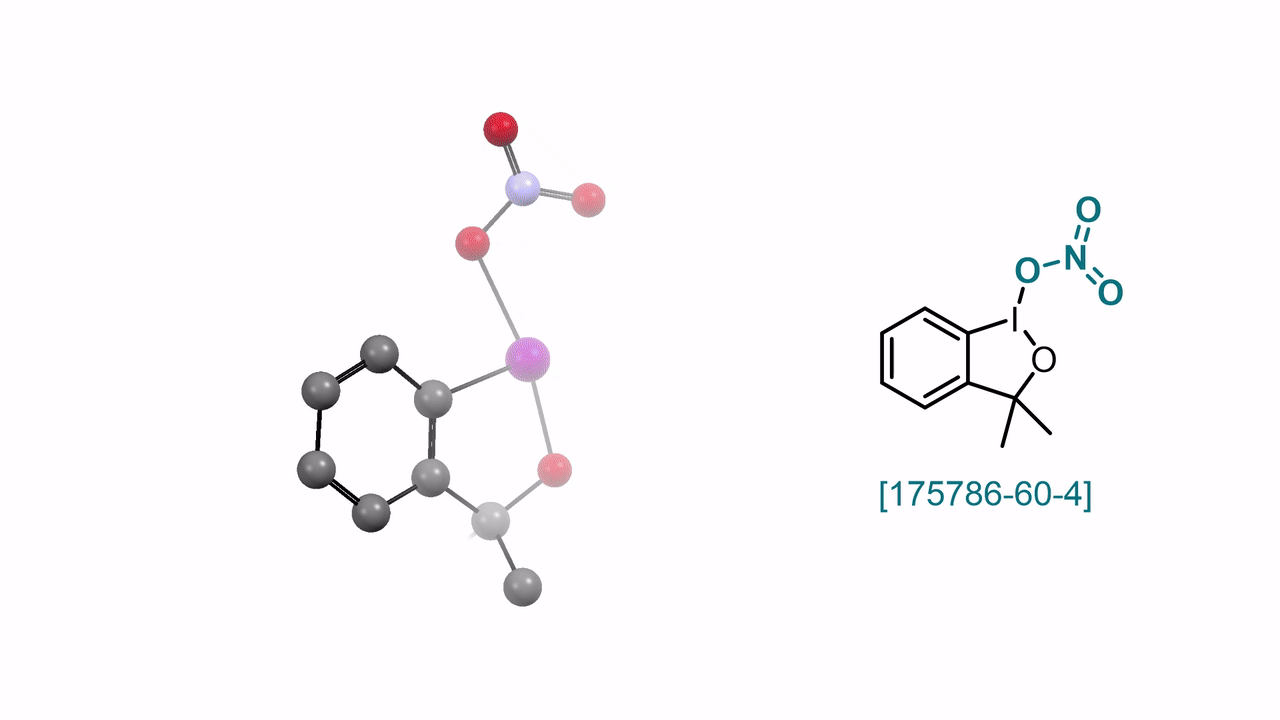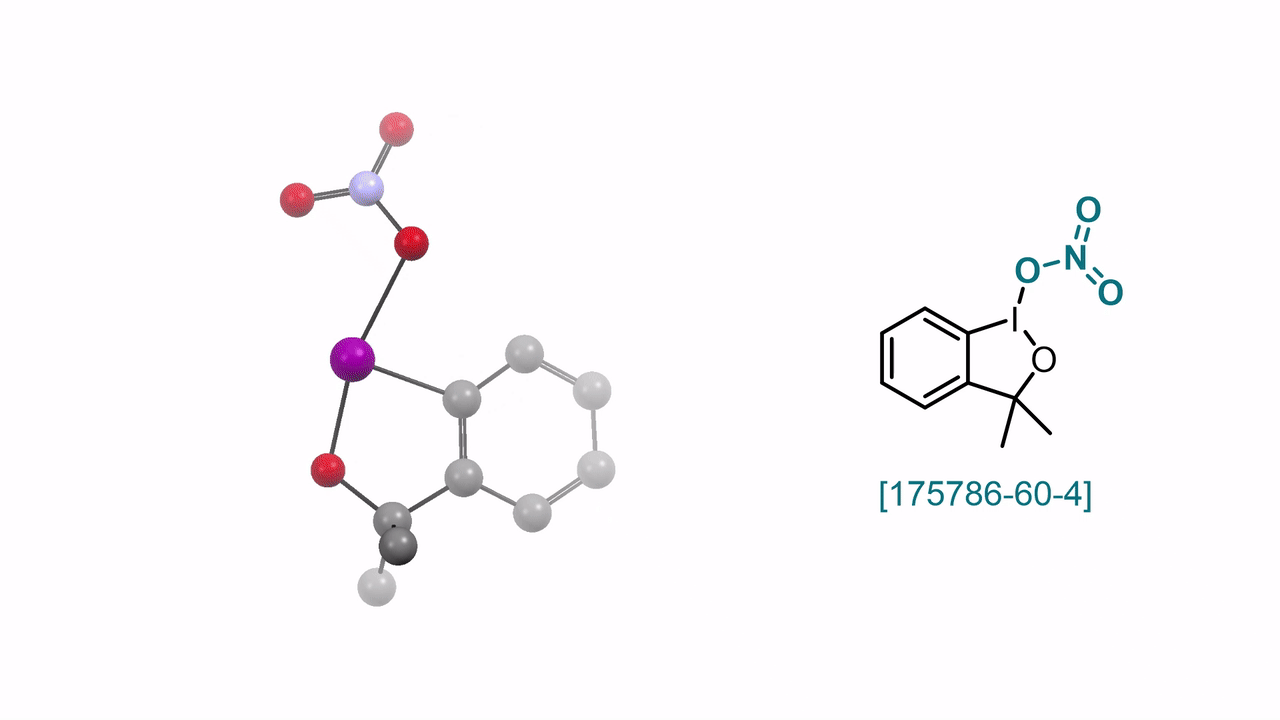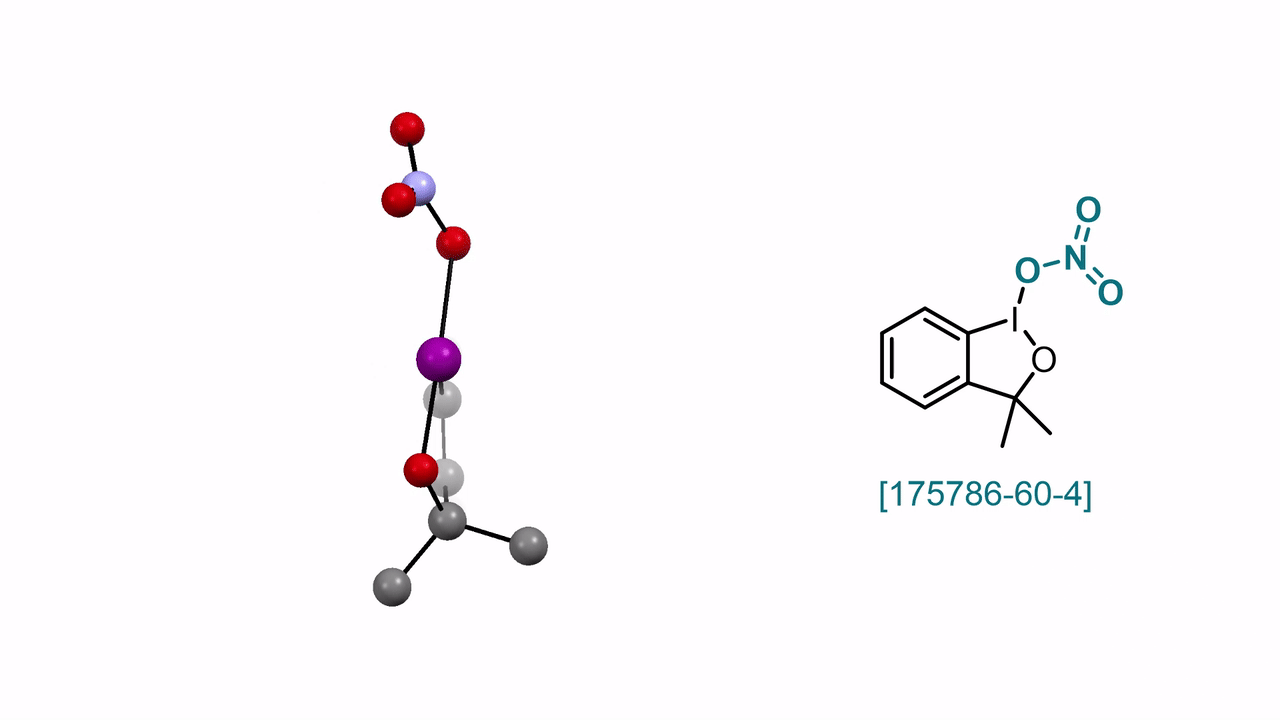
60. Encyclopedia of Reagents for Organic Synthesis (EROS 2025)
|
3,3-Dimethyl-1λ3-benzo[d][1,2]iodaoxol-1(3H)-yl Nitrate A. J. Fernandes, D. Katayev
|
59. Encyclopedia of Reagents for Organic Synthesis (EROS 2025)
|
N-Nitrosaccharin V. Valsamidou, S. Patra, D. Katayev
|
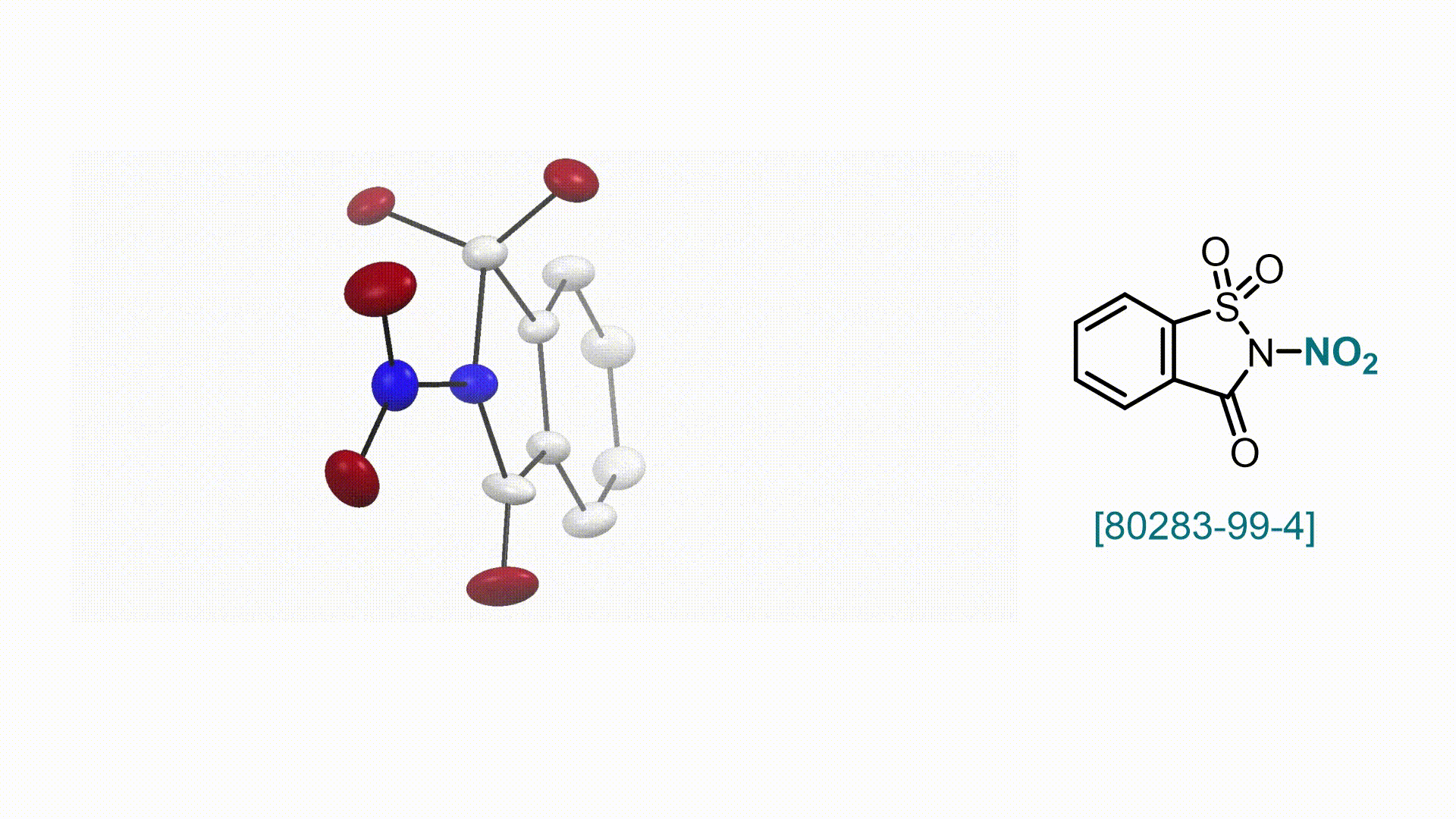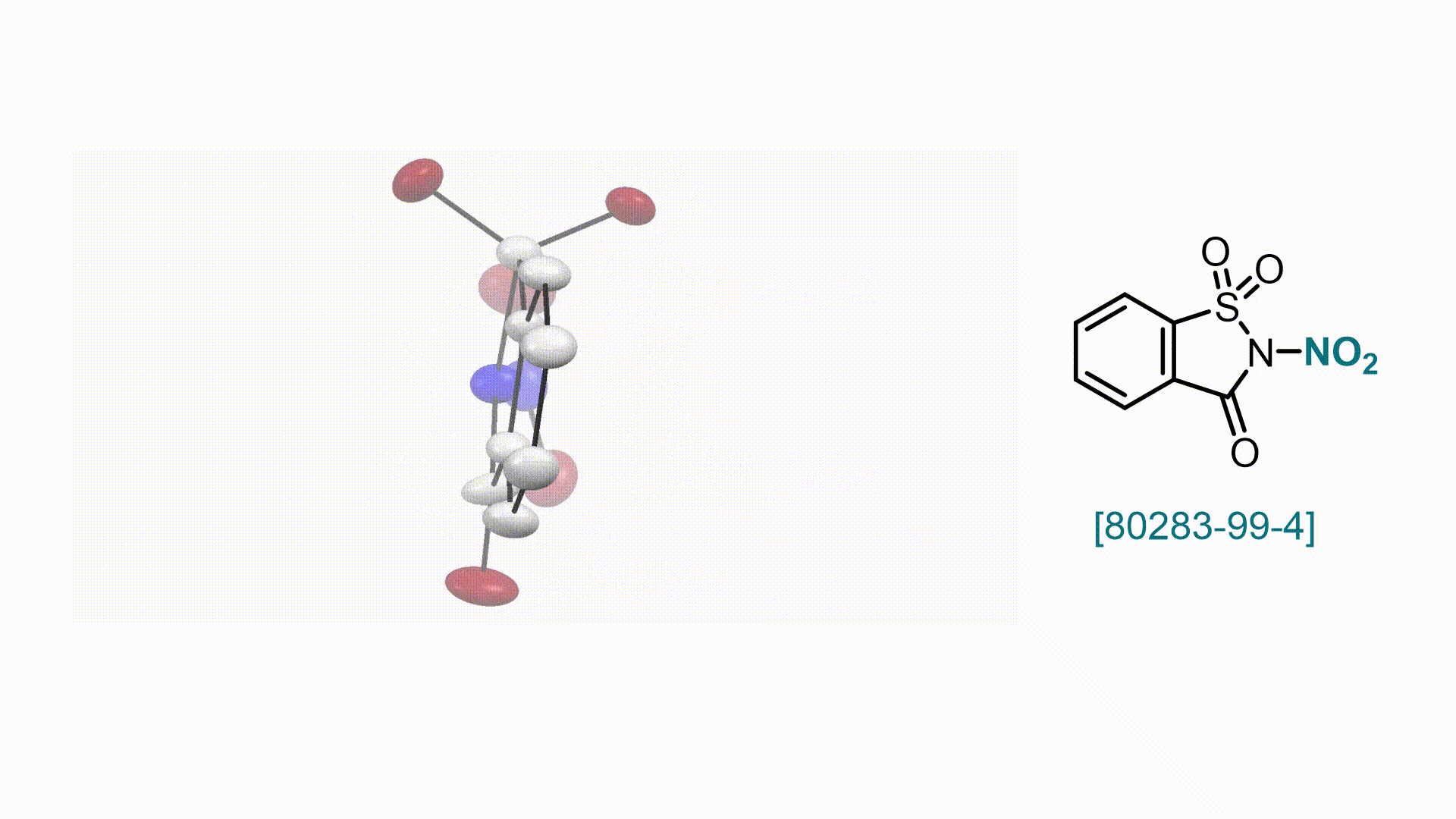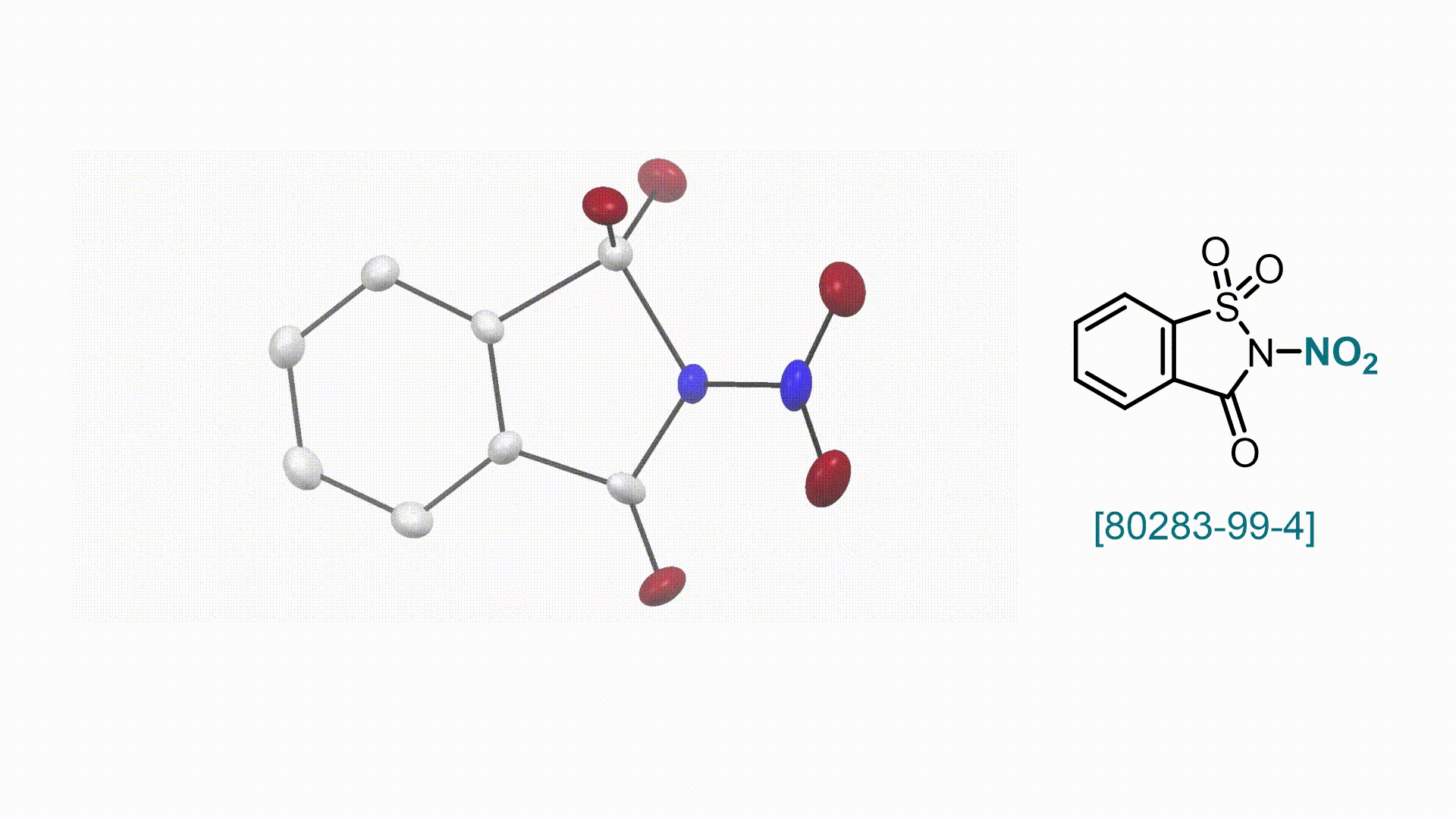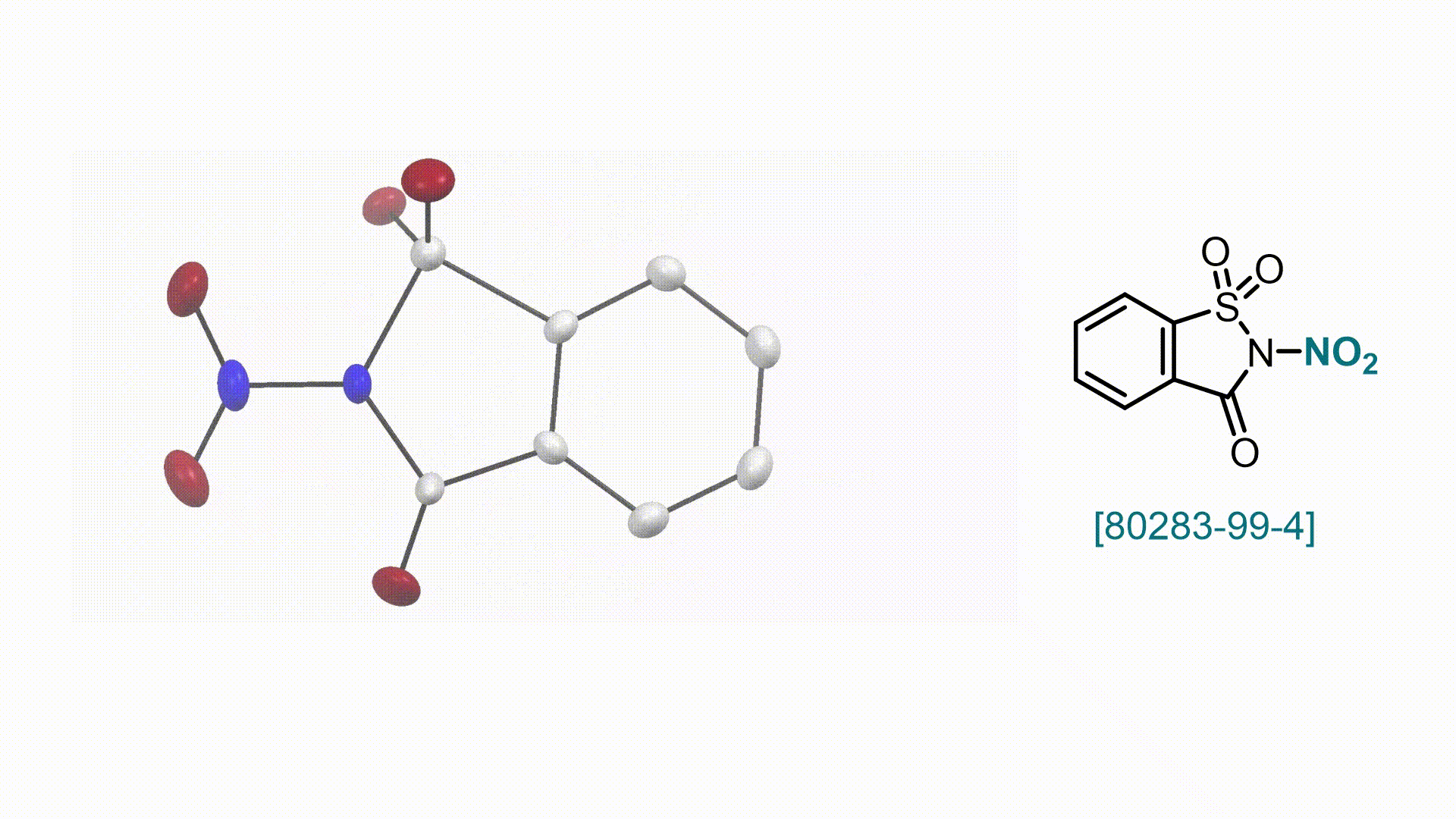
58. Encyclopedia of Reagents for Organic Synthesis (EROS 2025)
|
N-Nitrosuccinimide S. Patra, D. Katayev
|
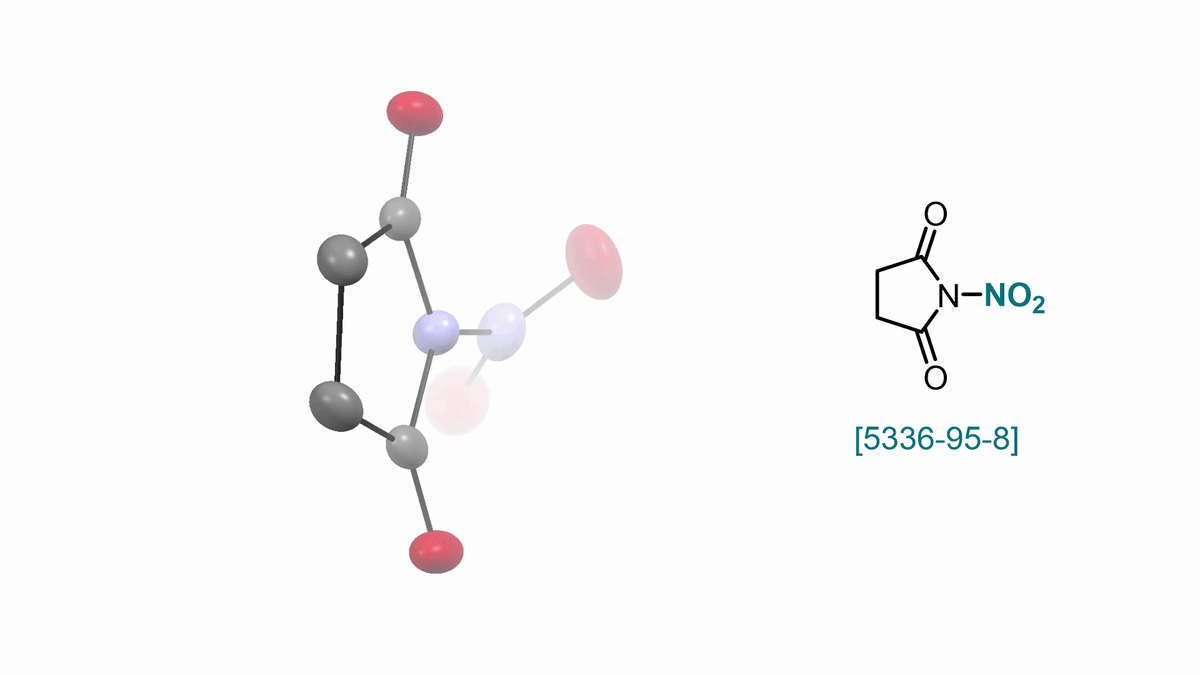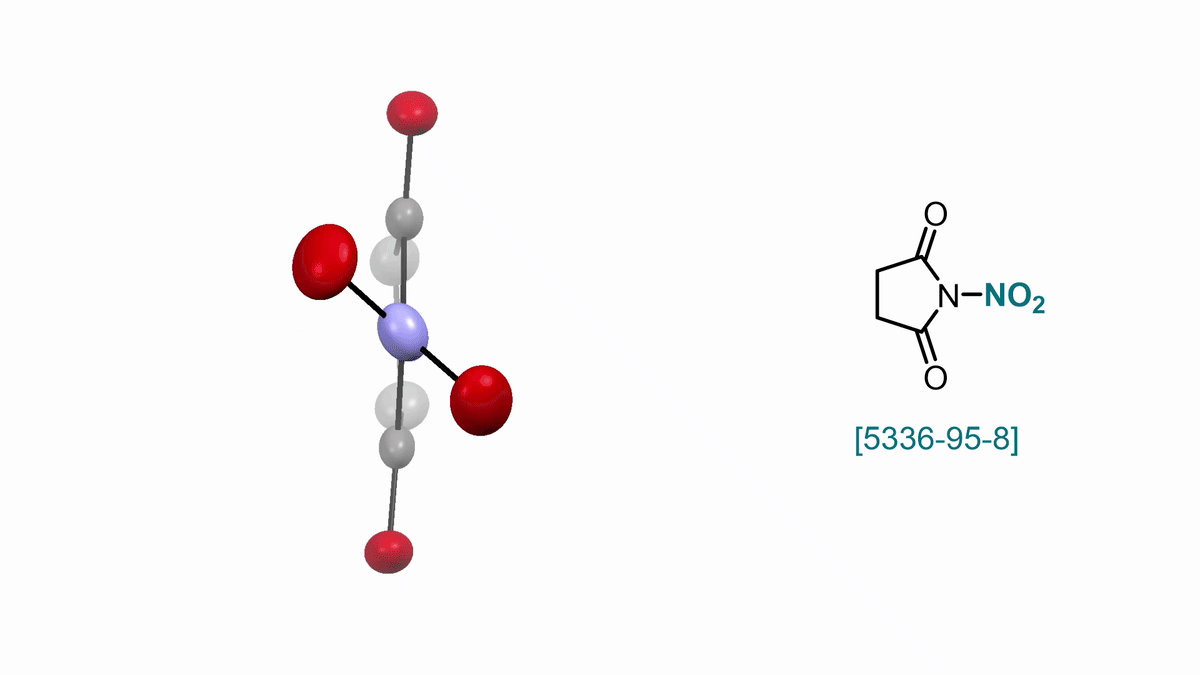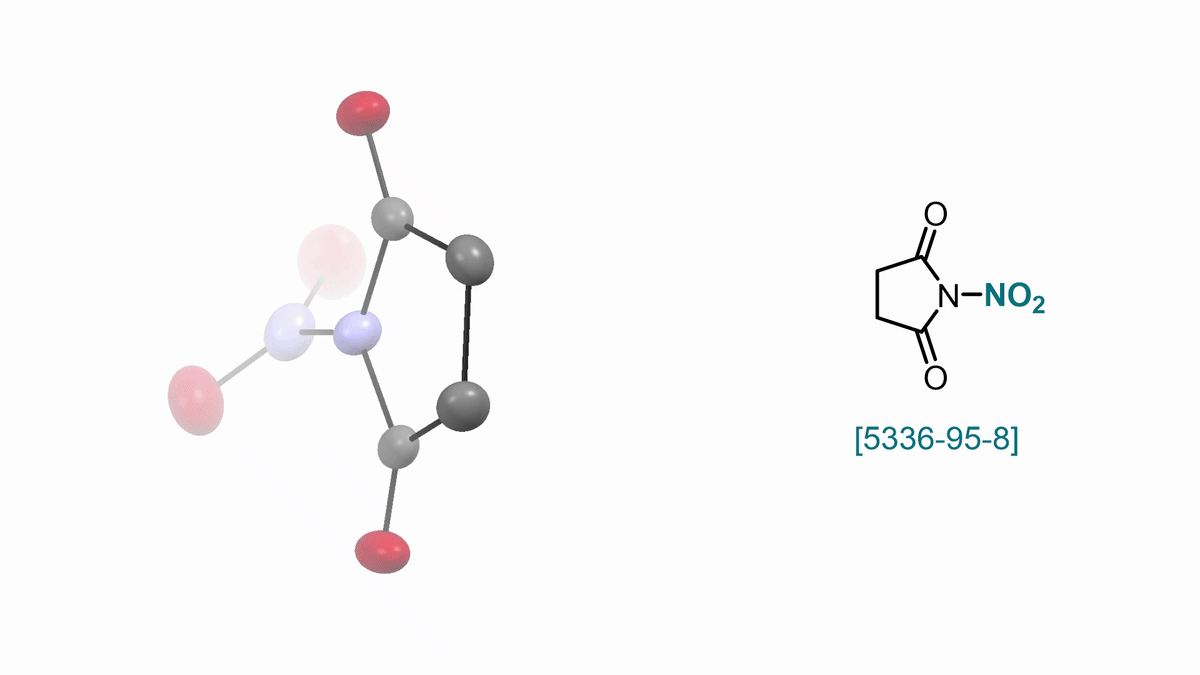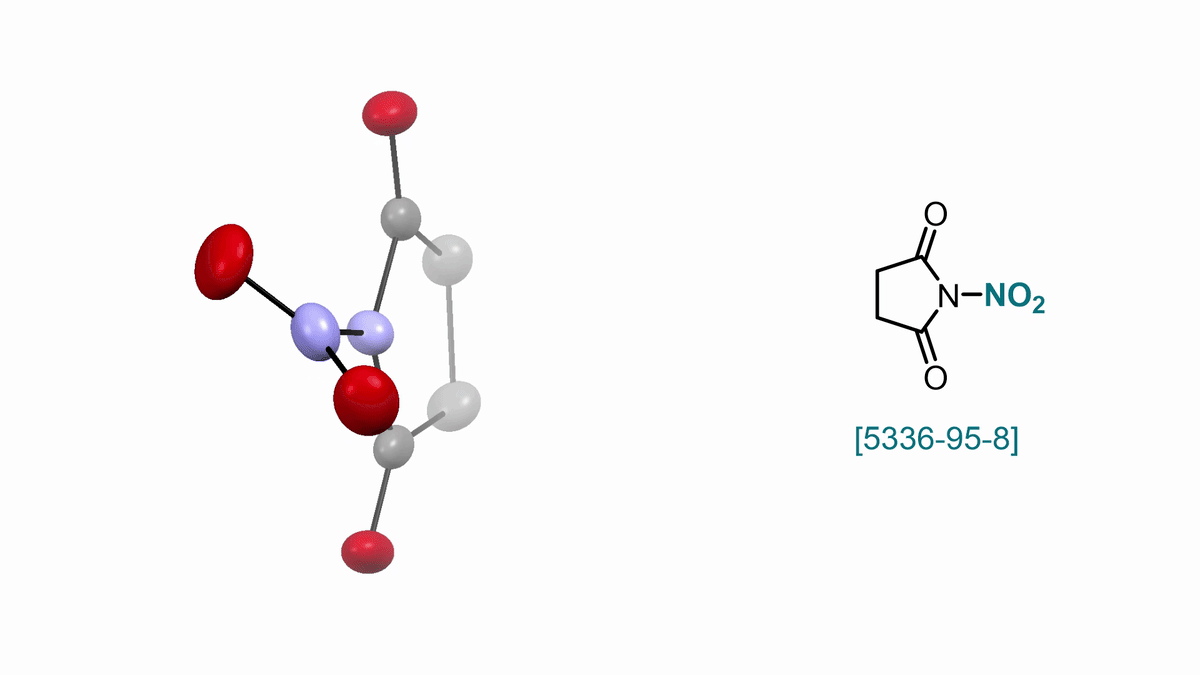
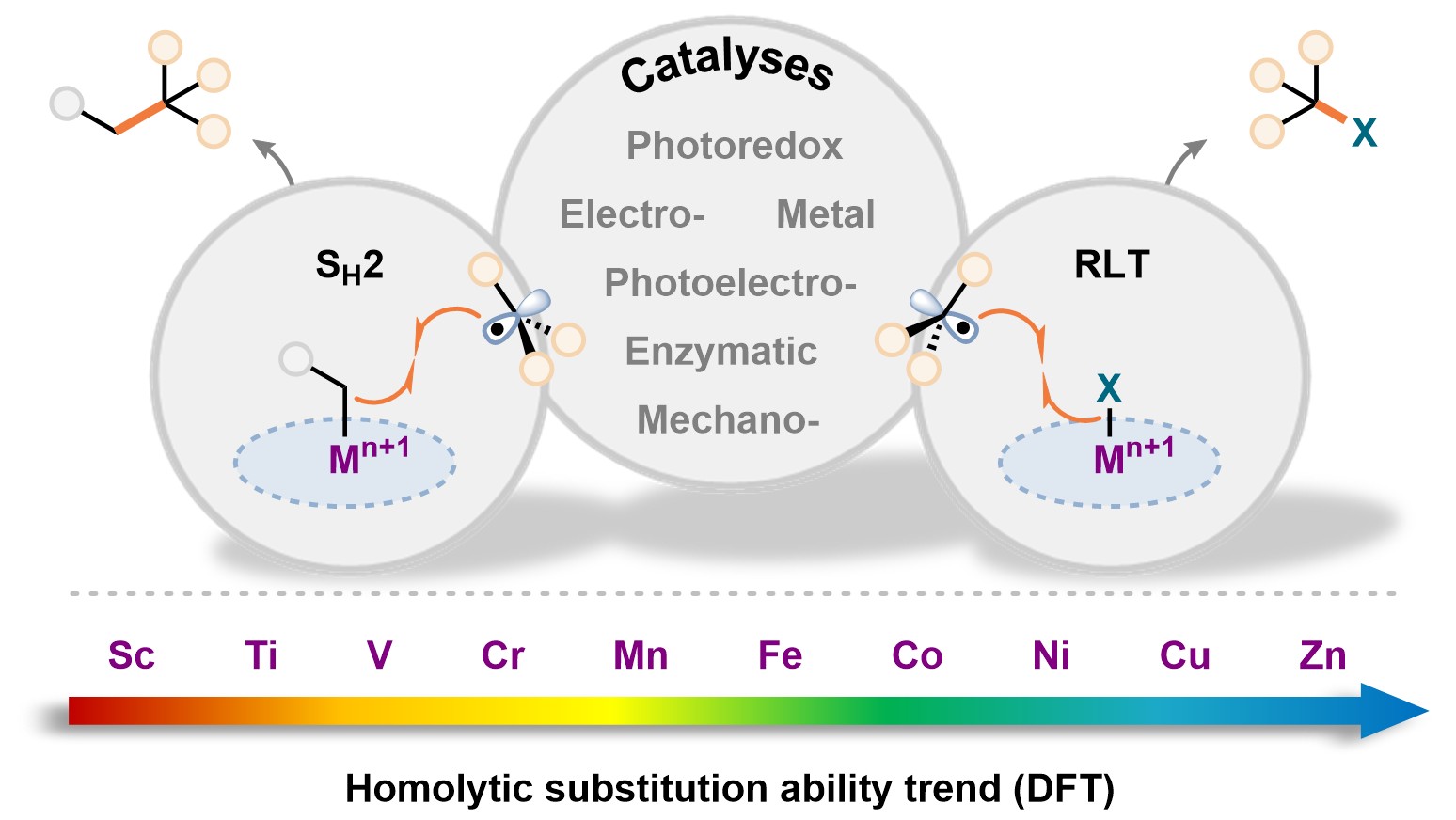
57. ACS Cent. Sci. 2025, 11, 10, 1812–1827
|
Bimolecular Homolytic Substitution (SH2) and Radical Ligand Transfer A. J. Fernandes, D. Katayev |
56. Angew. Chem. Int. Ed. 2025, 64, e202508377
|
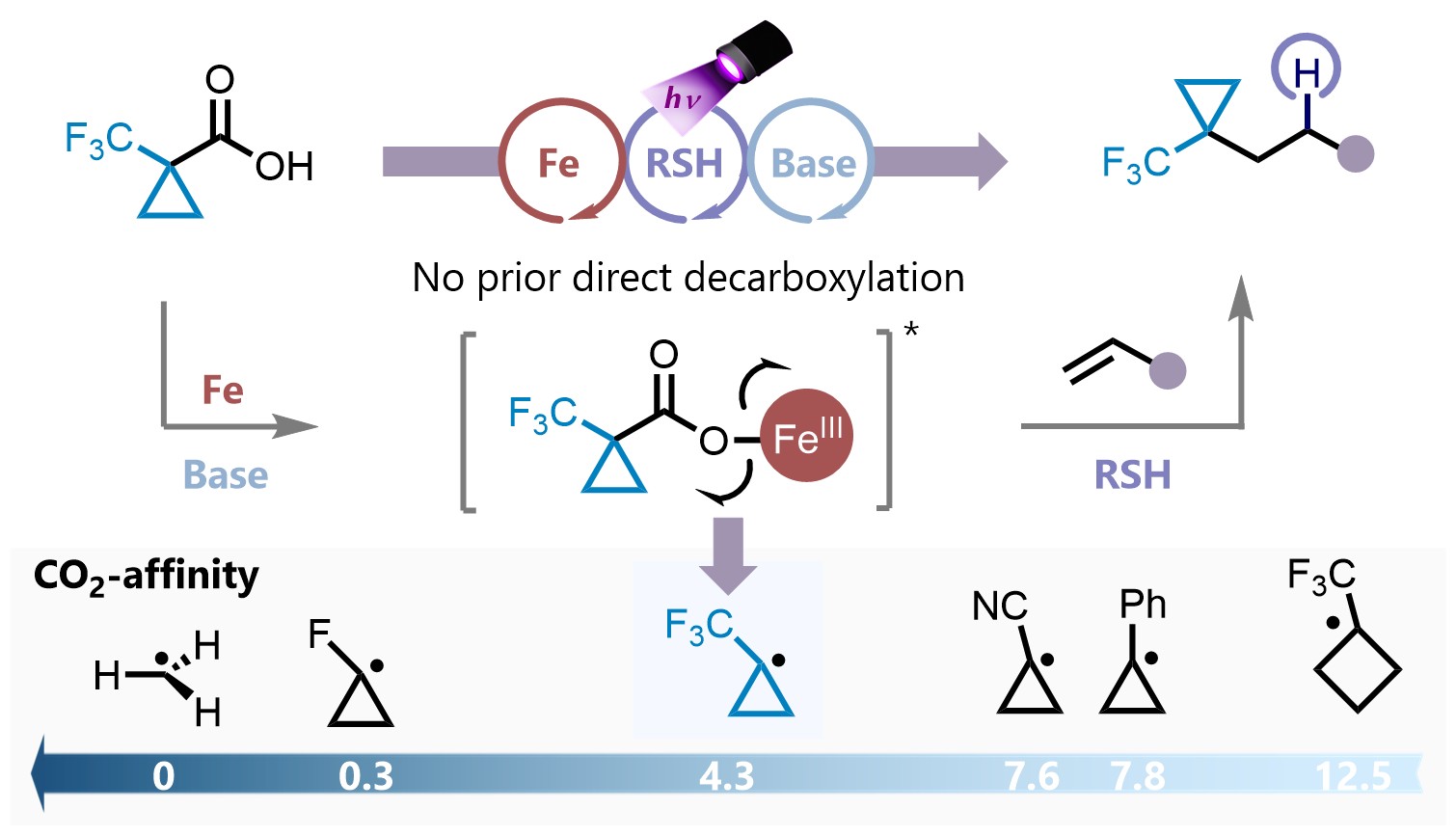
Overcoming Challenges in Small-Ring Transfer: Direct Decarboxylative Hydroalkylation of Alkenes via Iron-Thiol Catalysis R. Giri, P-K. Peng, A. J. Fernandes, S. Yu, J. G. West, D. Katayev |
55. Green Chem. 2025, 27, 7122-7128
|
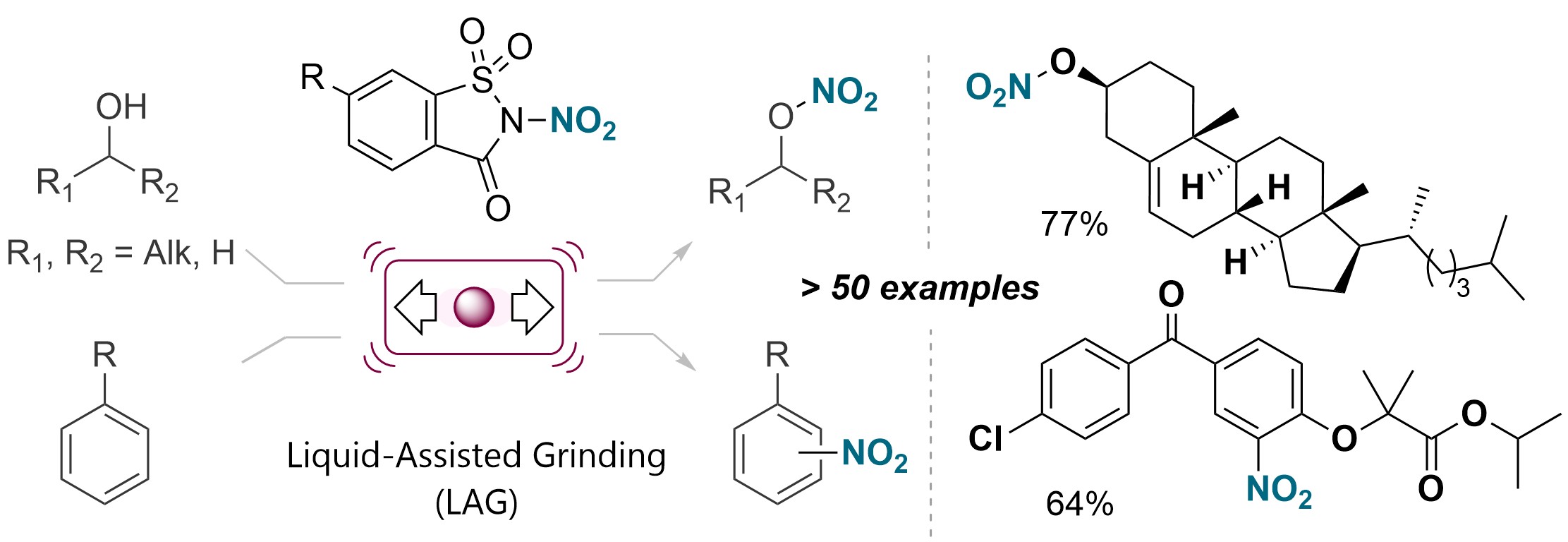
Mechanochemical Nitration of Arenes and Alcohols Using a Bench-Stable Organic Nitrating Reagent V. Valsamidou, S. Patra, B. Kadriu, M. G. Metzger, L. Gremaud, D. Katayev
|
54. Acc. Chem. Res. 2025, 58, 13, 2046–2060
|
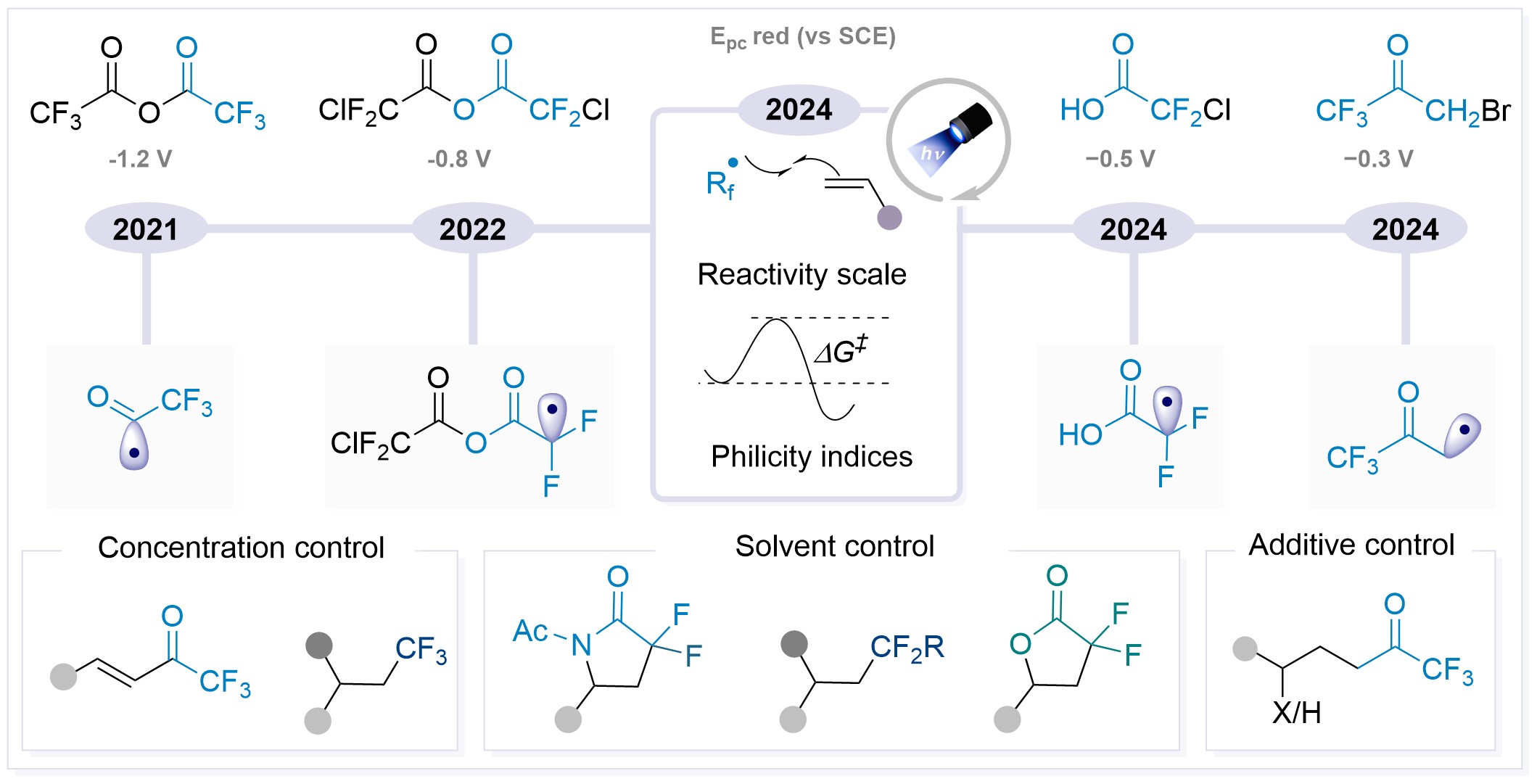
Fluorinated Radicals in Divergent Synthesis via Photoredox Catalysis R. Giri, A. J. Fernandes, D. Katayev
|
53. Synlett, Synpacts. 2025, 36, A-G
|
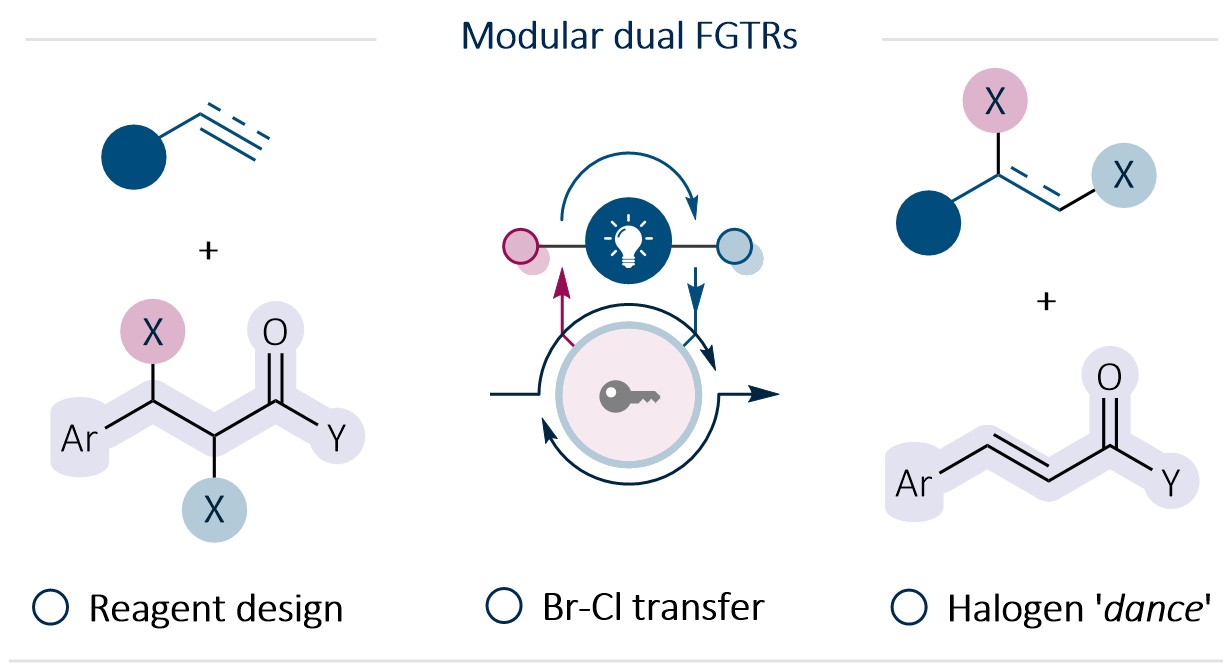
Dual Functional Group Transfer Reagents for Dihalogenation Reactions E. Zhilin, R. Giri, D. Katayev
|
52. Chem. Commun. 2025, 61, 1689-1692
|
Photoredox Radical/Polar Crossover Enables Carbo-Heterofunctionalization of Alkenes: Facile Access to 1,3-Difunctionalization Nitro Compounds S. Patra, V. Valsamidou, B. N. Nandasana, D. Katayev
|
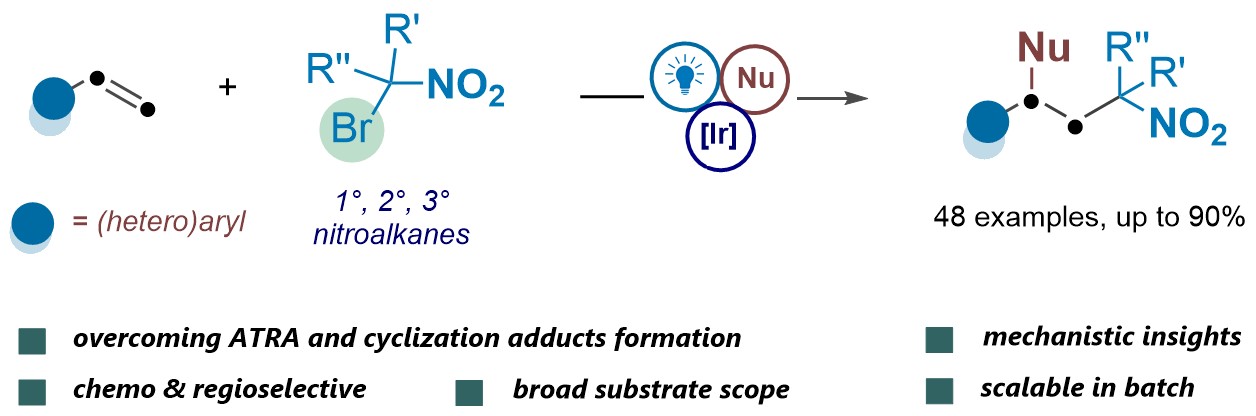
51. J. Am. Chem. Soc. 2024, 146, 31547−31559
|
Visible-Light-Mediated Vicinal Dihalogenation of Unsaturated C−C Bonds Using Dual-Functional Group Transfer Reagents R. Giri, E. Zhilin, M. Kissling, S. Patra, A. J. Fernandes, D. Katayev Top 10 most downloaded articles in JACS Nov. 2024 – Jan. 2025 Highlighted in SYNFACTS, 2025 (Open access)
|

50. Chem. Eur. J. 2024, 30, e202403654
|
Facile Access to Terminal Nitroalkanes via Anti-Markovnikov Hydronitration and Hydronitroalkylation of Alkenes Using Photoredox Catalysis Hot Topic Photocatalysis
S. Patra, D. Katayev (Open access)
|

49. Helv. Chim. Acta, 2024, 107, e202400125
|
Divergent Synthesis of Trifluoromethyl Ketones via Photoredox Activation of Halotrifluoroacetones R. Giri, M. Kissling, E. Zhilin, A. J. Fernandes, Q. E. L.Ordan, D. Katayev Dedicated to Prof. Erick Carreira, President of the 57th Bürgenstock-Conference 2024 Most Accessed article 2024 – 2025 (Open access)
|

48. ACS Catalysis, 2024, 14, 13747–13758
|
Merging Iron-Mediated Radical Ligand Transfer (RLT) Catalysis and Mechanochemistry for Facile Dihalogenation of Alkenes S. Patra, V. Valsamidou, B. N. Nandasana, D. Katayev
|

47. Angew. Chem. Int. Ed. 2024, 63, e202411073
|
Overcoming Challenges in O-Nitration: Selective Alcohol Nitration Deploying N,6-Dinitrosaccharin and Lewis Acid Catalysis A. J. Fernandes, V. Valsamidou, D. Katayev (Open access)
|
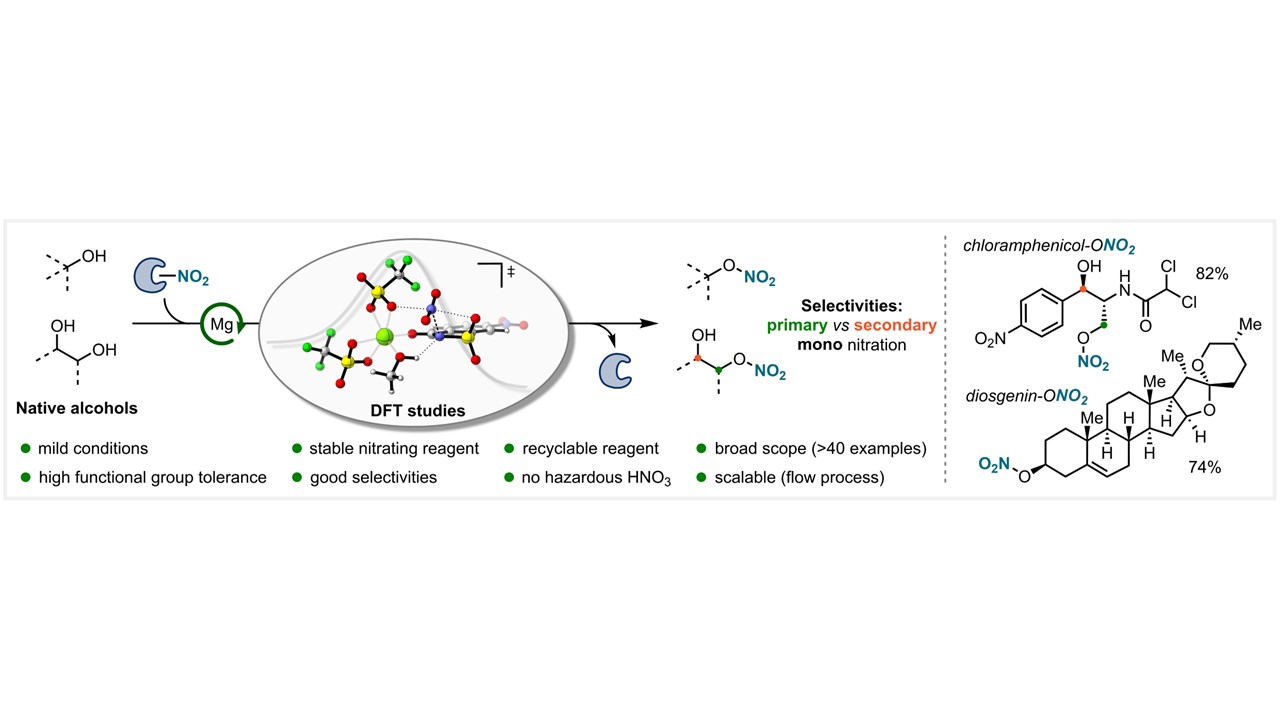
46. Chem. Sci. 2024, 15, 10659-10667
|
Divergent Functionalization of Alkenes Enabled by Photoredox Activation of CDFA and α-Halo Carboxylic Acids R. Giri, E. Zhilin, D. Katayev (Open access)
|
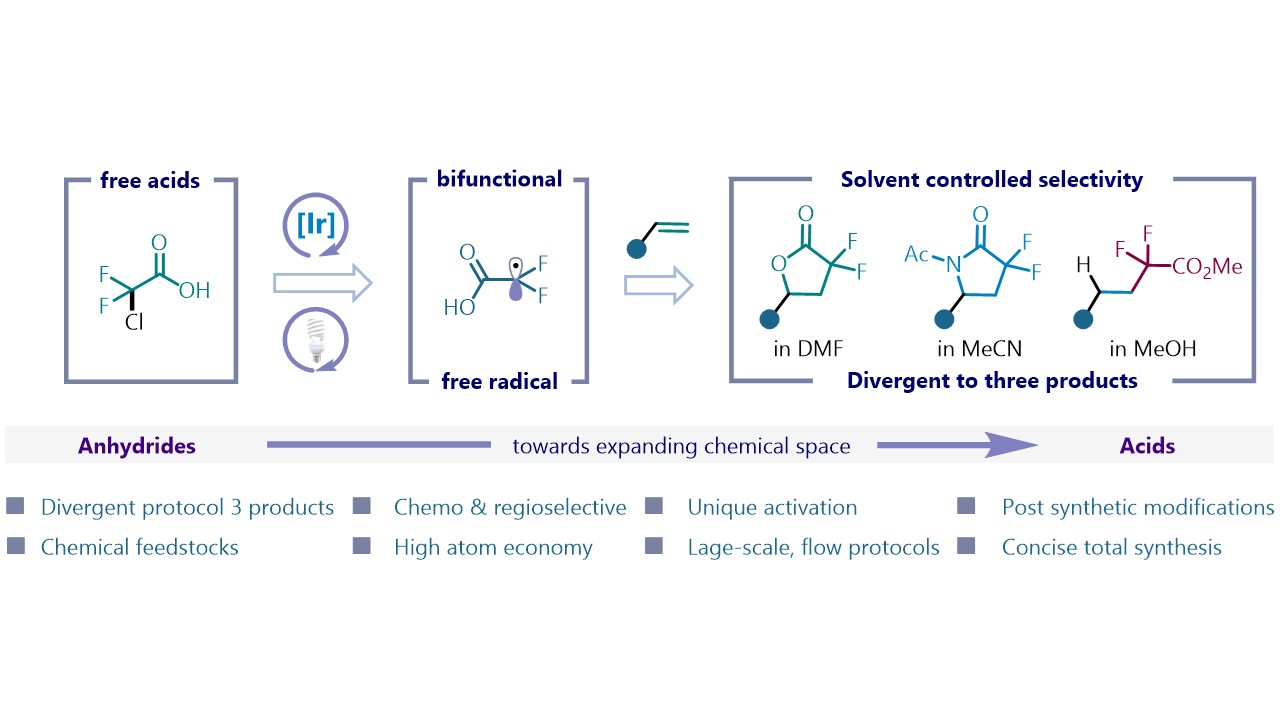
45. Adv. Sci. 2024, 11, 2402970
|
Mechanochemistry Drives Alkene Difunctionalization via Radical Ligand Transfer and Electron Catalysis S. Patra, B. N. Nandasana, V. Valsamidou, D. Katayev Highlighted in Chimia, 2024 Molecule of the month August 2024 DCBP
(Open access)
|

44. Angew. Chem. Int. Ed. 2024, 63, e202318377
|
Review and Theoretical Analysis of Fluorinated Radicals in Direct A. J. Fernandes, R. Giri, K. N. Houk, D. Katayev Appeared in Hot Topic: Fluorine Chemistry (Wiley) (Open access)
|

43. Chimia 2024, 78, 32
|
Simplifying Nitration Chemistry with Bench-stable Organic Nitrating Reagents S. Patra, V. Valsamidou, D. Katayev This review is a part of scientific contributions from ambizione fellows and young professors in chemistry from Switzerland (Open access)
|

42. ACS Catal. 2023, 13, 16136−16147
|
Nitrative Difunctionalization of Alkenes via Cobalt-Mediated Radical Ligand Transfer and Radical-Polar Crossover Photoredox Catalysis S. Patra, R. Giri, D. Katayev Highlighted in SYNFACTS, 2024
|
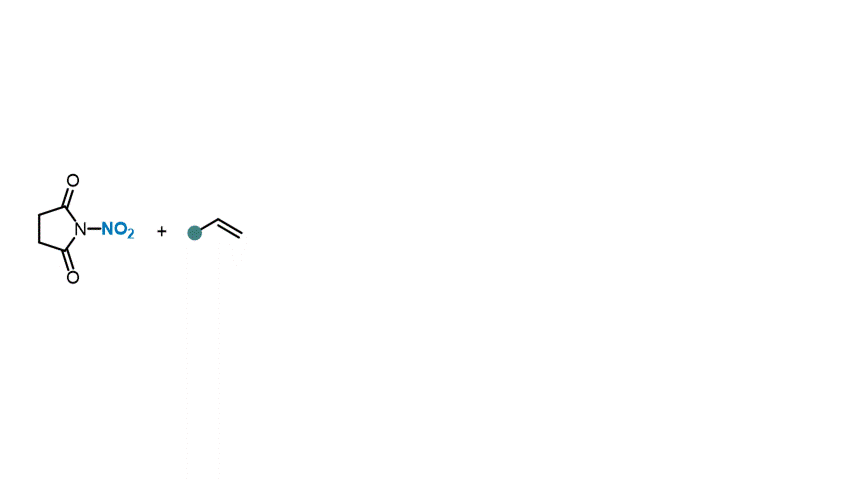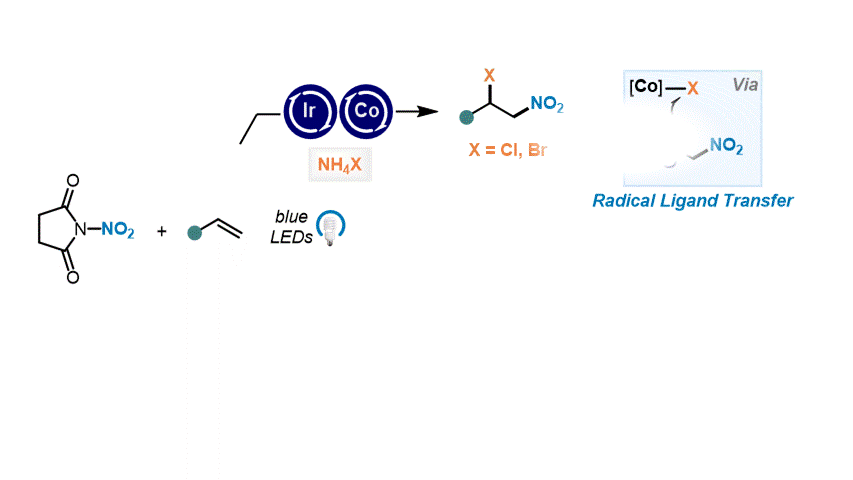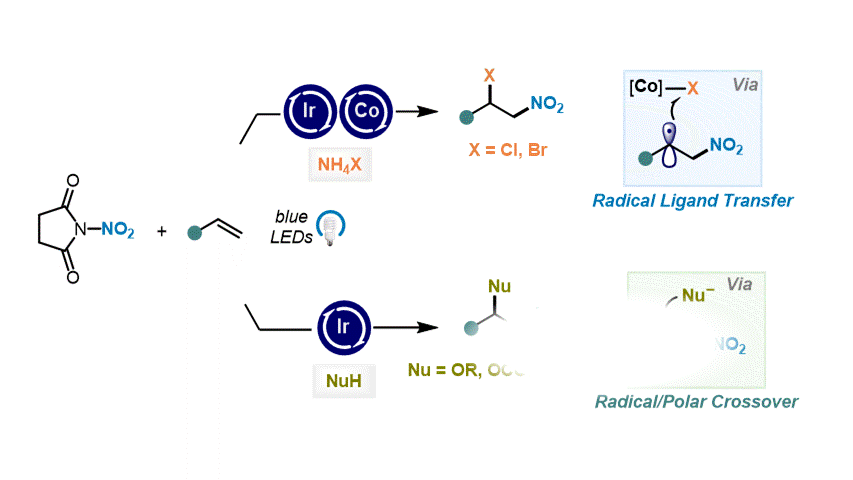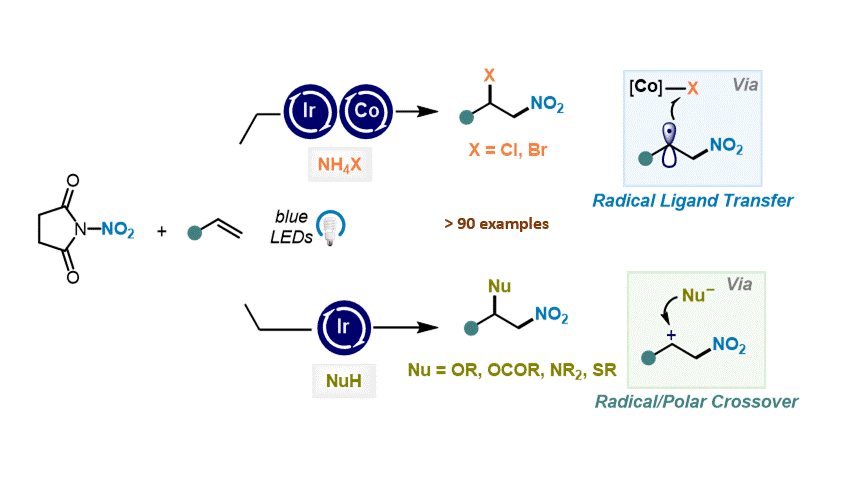
41. Angew. Chem. Int. Ed. 2023, 62, e202310851
|
Catalytic ipso-Nitration of Organosilanes Enabled by Electrophilic N-Nitrosaccharin Reagent I. Mosiagin, A. J. Fernandes, A. Budinska, L. Hayriyan, K. E. O. Ylijoki, D. Katayev German version: Angew. Chem. 2023, 135, e202310851 Highlighted in ChemistryViews, 2023 Highlighted in SYNFACTS, 2024 (Open access)
|

40. Angew. Chem. Int. Ed. 2023, 62, e202300533
|
Electron-Driven Nitration of Unsaturated Hydrocarbons S. Patra, I. Mosiagin, R. Giri, T. Nauser, D. Katayev German version : Angew. Chem. 2023, 135, e202300533 Highlighted in Chimia 2023, 77, 534 (Open access)
|
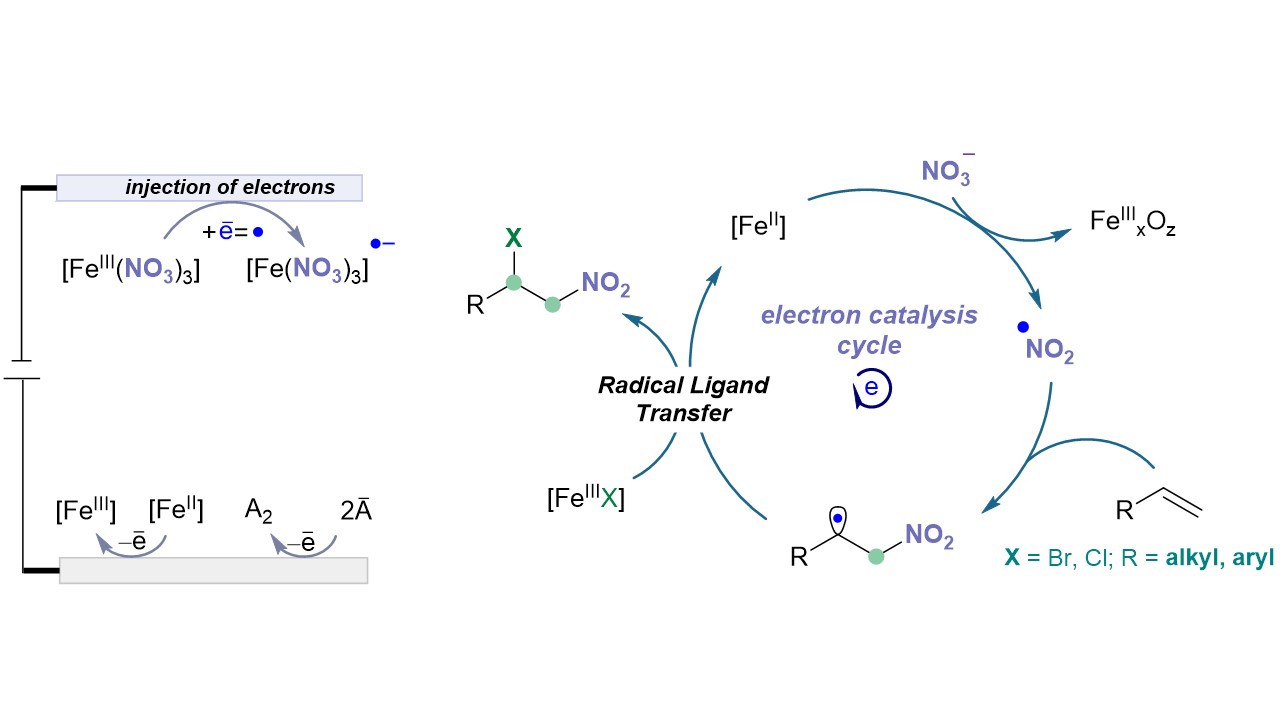
39. ChemCatChem, 2023, 15, e202201427
|
Nitryl Radical-Triggered Semipinacol-Type Rearrangement, Lactonization, and Cycloetherification of Olefins R. Giri, S. Patra, D. Katayev Invited Article: This publication is part of a Special Collection on “Photocatalytic Synthesis”. Please check the ChemCatChem homepage for more articles in the collection. (Open access)
|
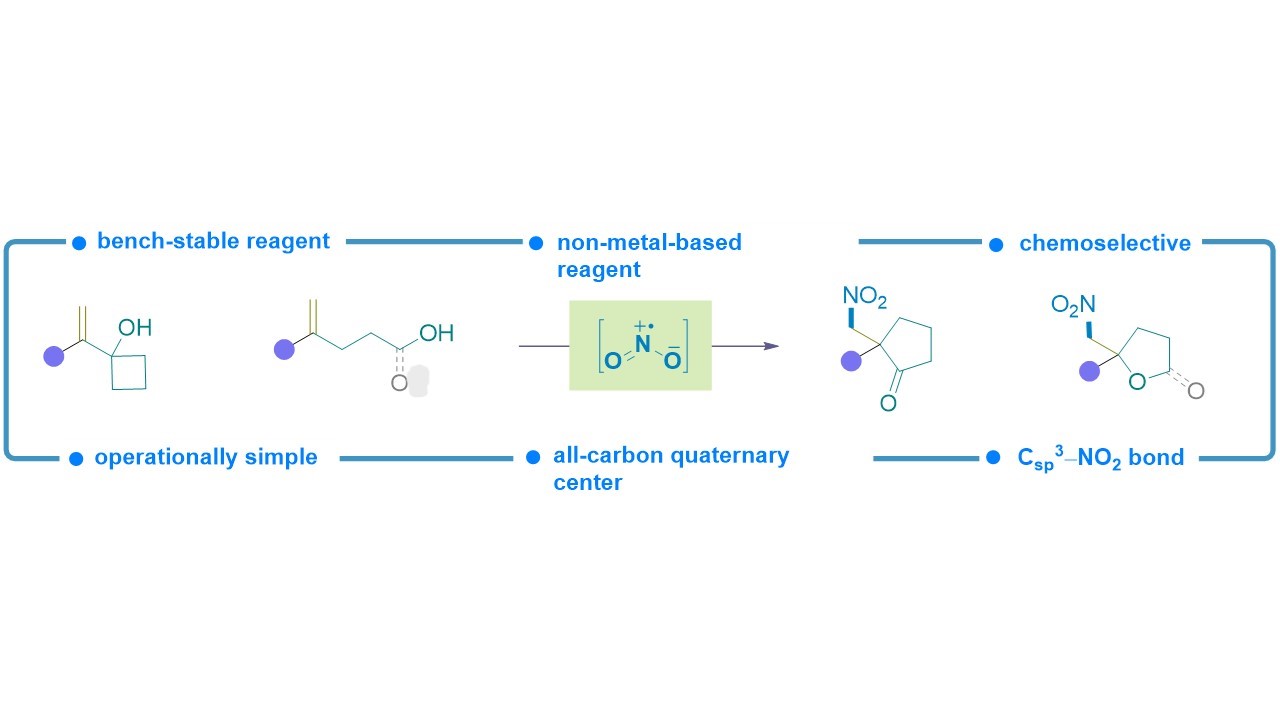
38. Angew. Chem. Int. Ed. 2022, 61, e202209143
|
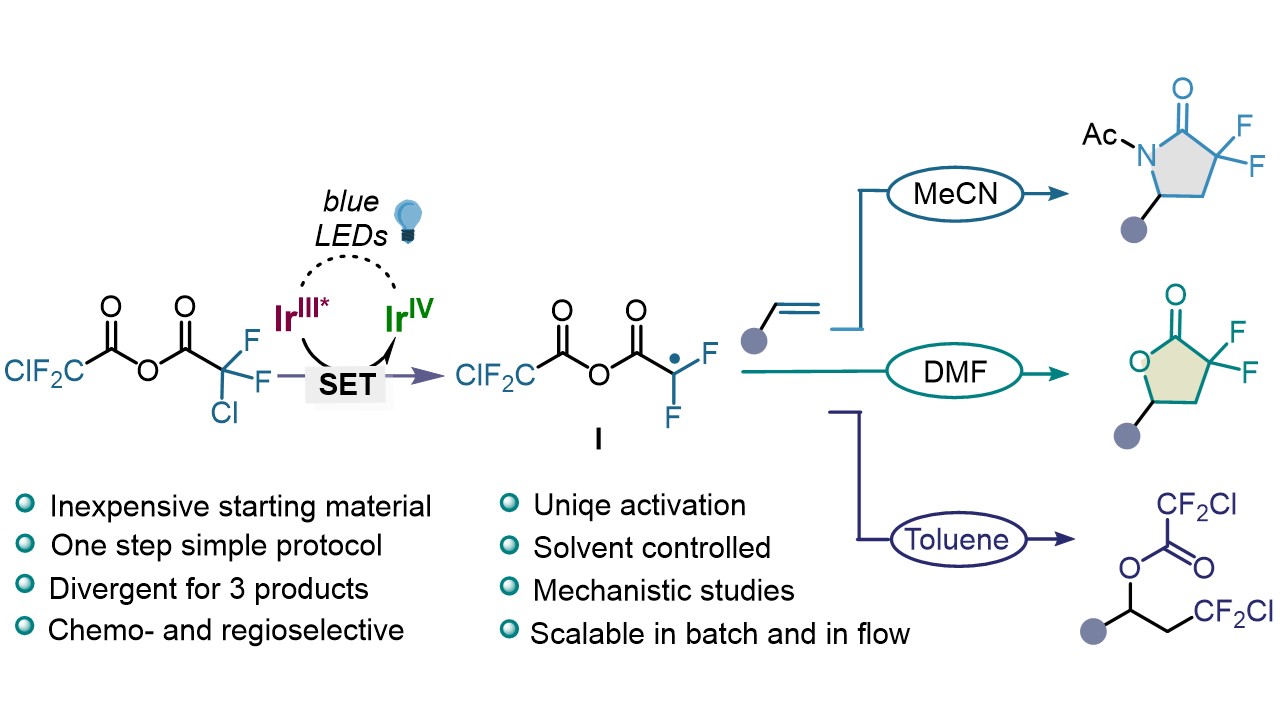
Solvent-Controlled Switchable Synthesis of gem-Difluoro Compounds from Anhydrides under Photoredox Catalysis R. Giri, I. Mosiagin, I. Franzoni, N. Y. Nötel, S. Patra, D. Katayev German version : Angew. Chem. 2022, 134, 202209143 Preprint : ChemRxiv Top downloaded article of 2022 in Angew Chem (Open access)
|
37. Synthesis 2022, 54, 3432–3472
|
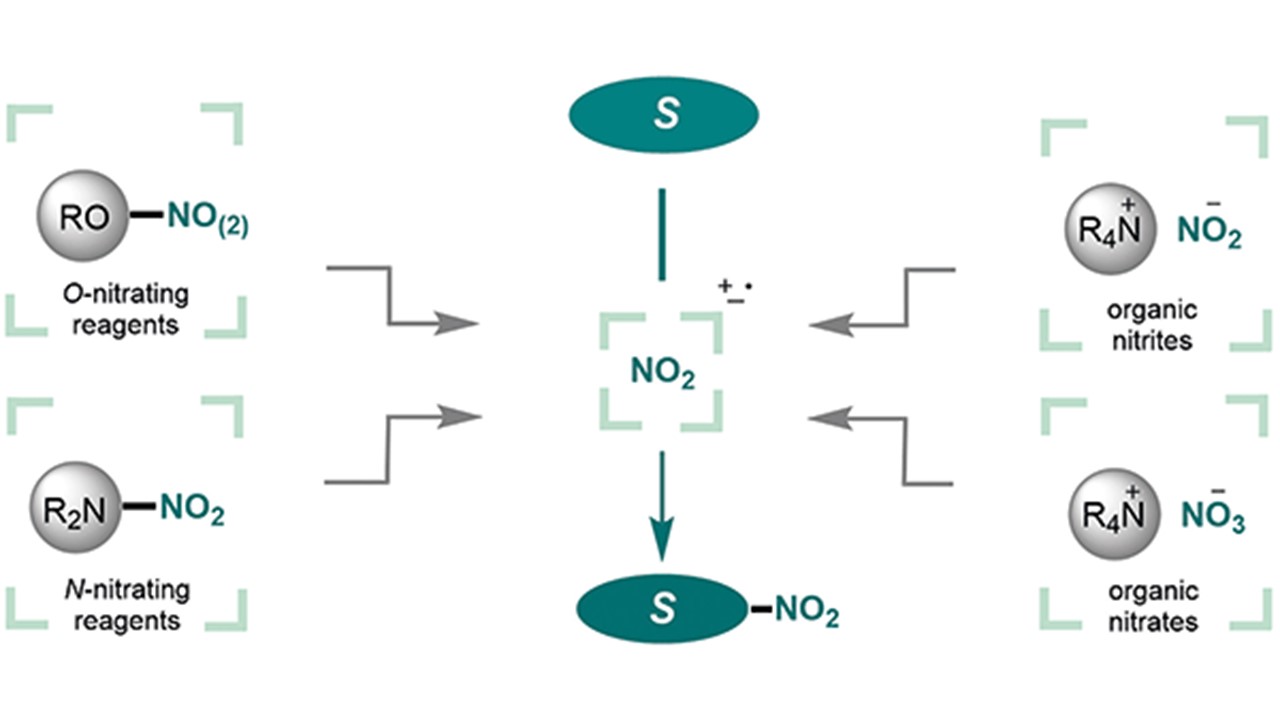
Organic Nitrating Reagents S. Patra, I. Mosiagin, R. Giri, D. Katayev Invited Review by Prof. Paul Knochel; Bürgenstock Special Section 2021 – Future Stars in Organic Chemistry (Open access)
|
36. Angew. Chem. Int. Ed. 2021, 60, 22487-22495
|
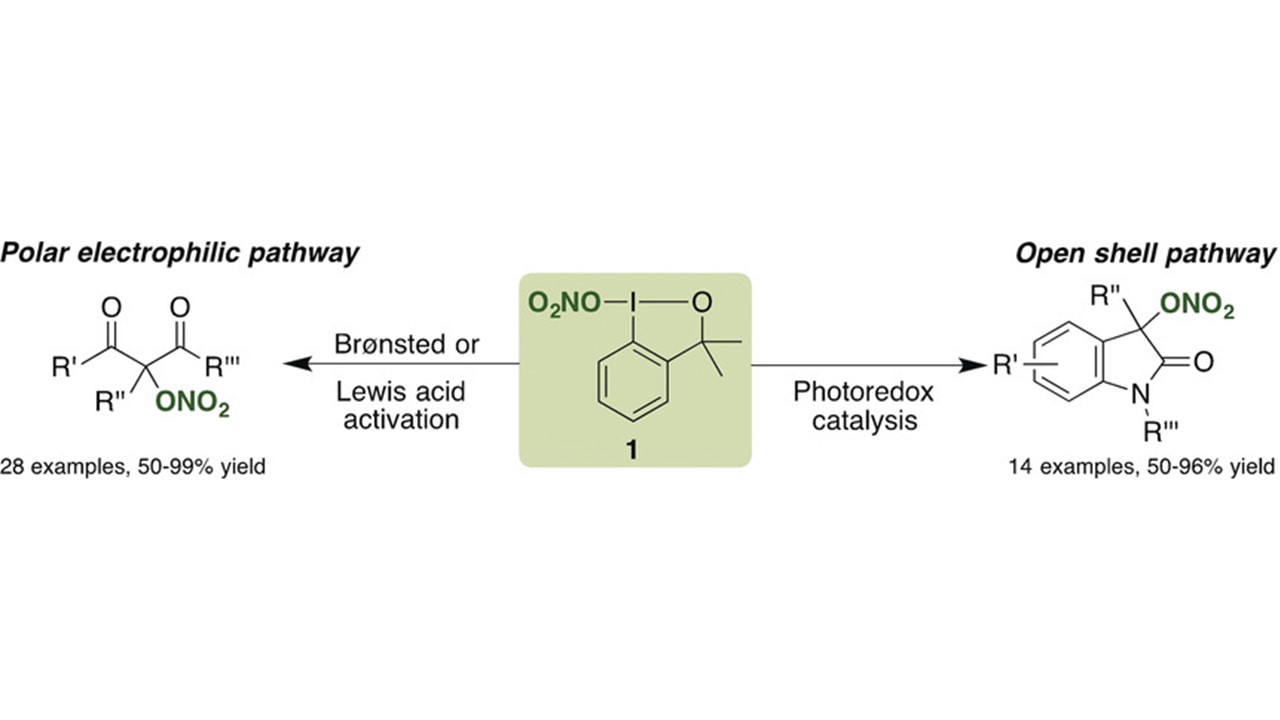
Radical Trifluoroacetylation of Alkenes Triggered by a Visible-Light-Promoted C-O Bond Fragmentation of Trifluoroacetic Anhydride K. Zhang, D. Rombach, N. Y. Nötel, G. Jeschke, D. Katayev German version: Angew. Chem. 2021, 133, 22661–22669 Highlighted in SYNFACTS, 2021 Dedicated to Prof. Peter Kündig on the occasion of his 75th birthday (Open access)
|

35. Helv. Chim. Acta 2021, 104, e2000193
|
Asymmetric Synthesis of Perfluoroalkylated α-Amino Acids through Generated Radicals Using a Chiral Ni(II) Complex N. V. Stoletova, A. D. Moshchenkov, A. F. Smol’yakov, Z. T. Gugkaeva, V. I. Maleev, D. Katayev, V. A. Larionov Dedicated to our friend and colleague Prof. Antonio Togni on the occasion of his 65th birthday (Open access)
|

34. Angew. Chem. Int. Ed. 2020, 59, 17162-17168
|
Synthesis, Characterization, and Diverse Reactivity of a Hypervalent Iodine‐Based Nitrooxylating Reagent R. Calvo, A. Tellier, T. Nauser, D. Rombach, D. Nater, D. Katayev German Version: Angew. Chem. 2020, 132, 17312–17319 Dedicated to Professor Antonio Togni on the occasion of his 65th birthday (Open access)
|
33. Org. Lett. 2020, 22, 2714-2719
|
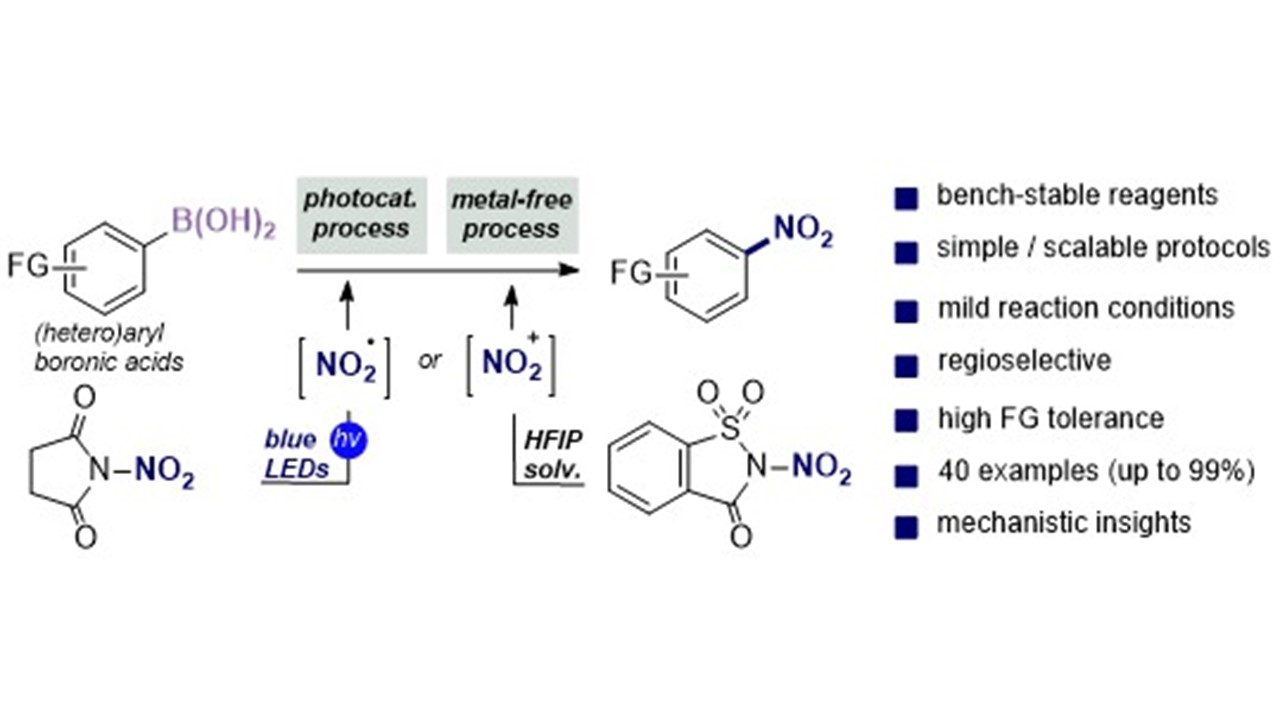
N-Nitroheterocycles: Bench-Stable Organic Reagents for Catalytic Ipso-Nitration of Aryl and Heteroarylboronic Acids K. Zhang, A. Budinská, A. Passera, D. Katayev Highlighted in Org. Process. Res. Dev. 2020, 24, 874-883 (Open access)
|
32. Angew. Chem. Int. Ed. 2020, 59, 2149
|
Bunsen Lecture: R. Winter/Werner Prize: Konrad Tiefenbacher/Ružička Prize D. Katayev (Open access)
|

31. The Catalyst Review, 2020, 33, 1-7
|
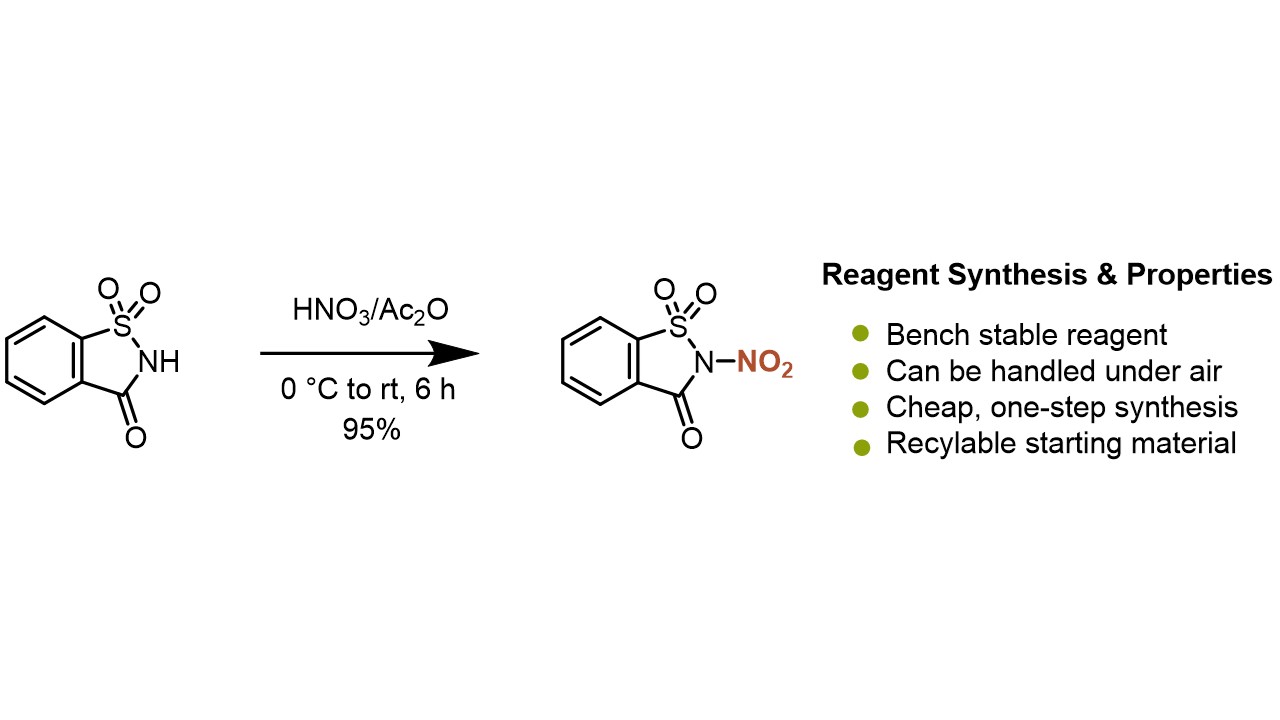
Simplifying Nitration Chemistry with a Bench-stable and Easily Accessible Reagent R. Calvo, D. Katayev (Open access)
|

30. Patent: 2019, WO2020084059A1
|
Patent: “N-Nitrosaccharins” D. Katayev, K. Zhang, R. Calvo (Open access)
|
29. Patent: 2019, EP3717464A1
|
Patent: “Nitration” D. Katayev, K. Zhang, R. Calvo (Open access)
|

28. Chem. Eur. J. 2019, 25, 12929-12939
|
Synthetic Diversity from a Versatile and Radical Nitrating Reagent K. Zhang, B. Jelier, A. Passera, G. Jeschke, D. Katayev Published as front page cover
(Open access)
|
27. Nat. Commun. 2019, 10, 3410-3418
|
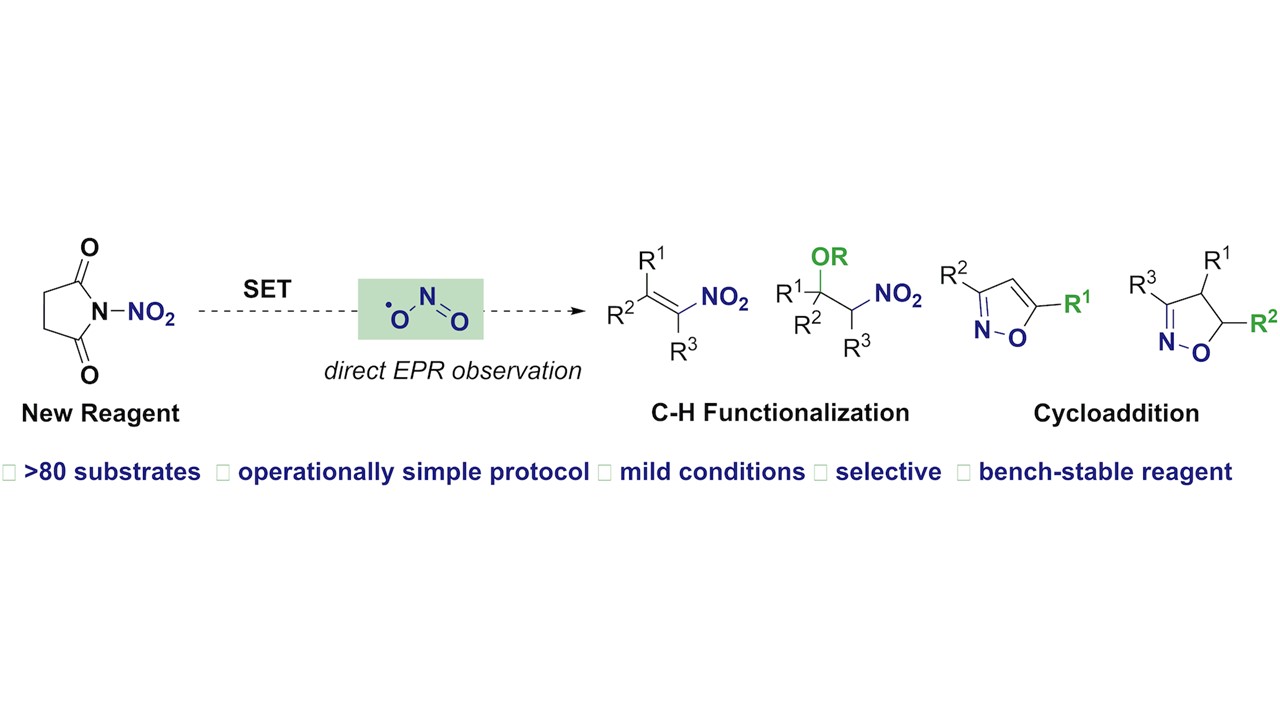
Facile Access to Nitro(hetero)arenes Using N-Nitrosacchrain: An Electrophilic and Shelf-Stable Nitrating Reagent R. Calvo, K. Zhang, A. Passera, D. Katayev Highlighted in Springer Nature, 2019 (Open access)
|
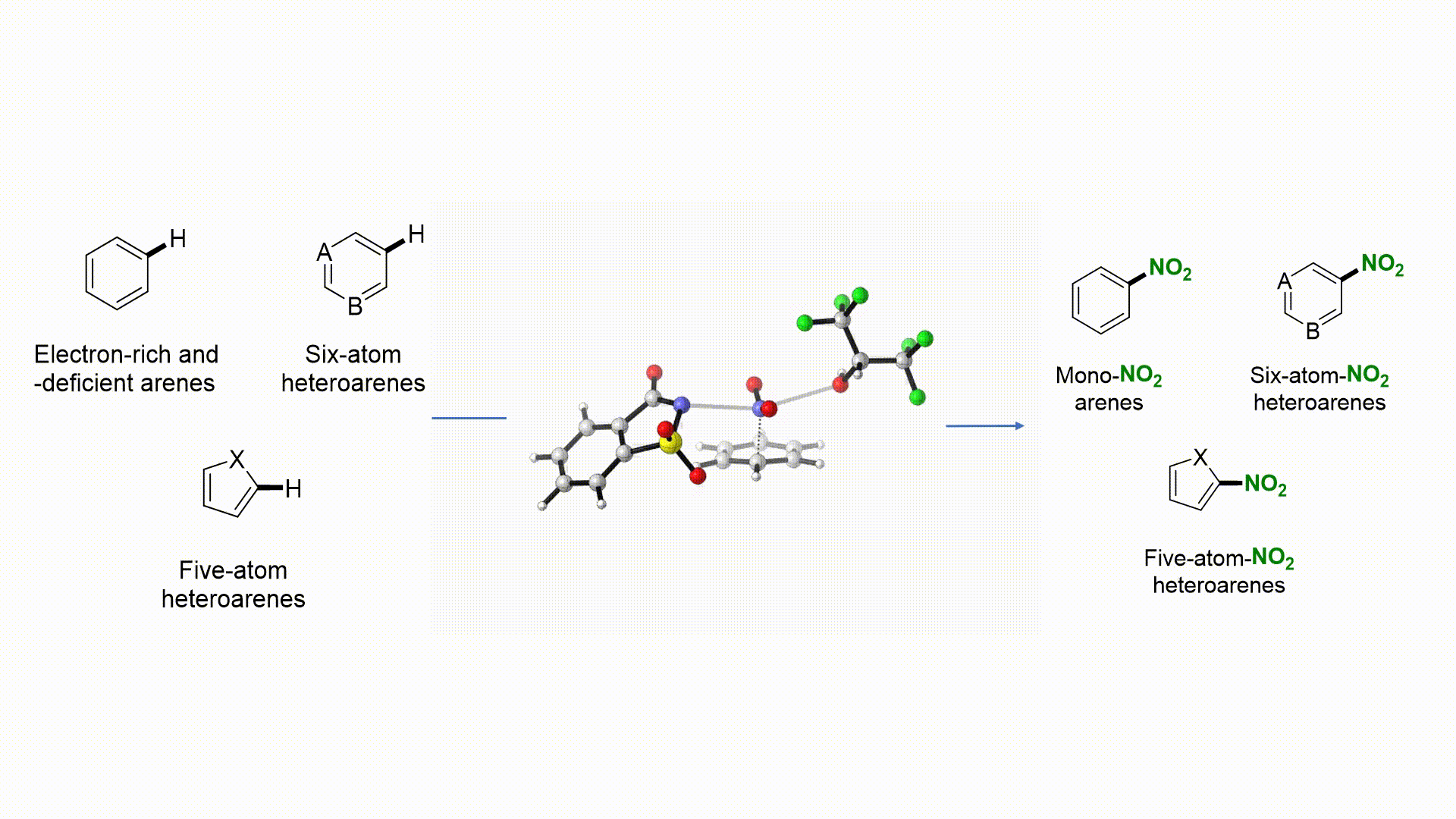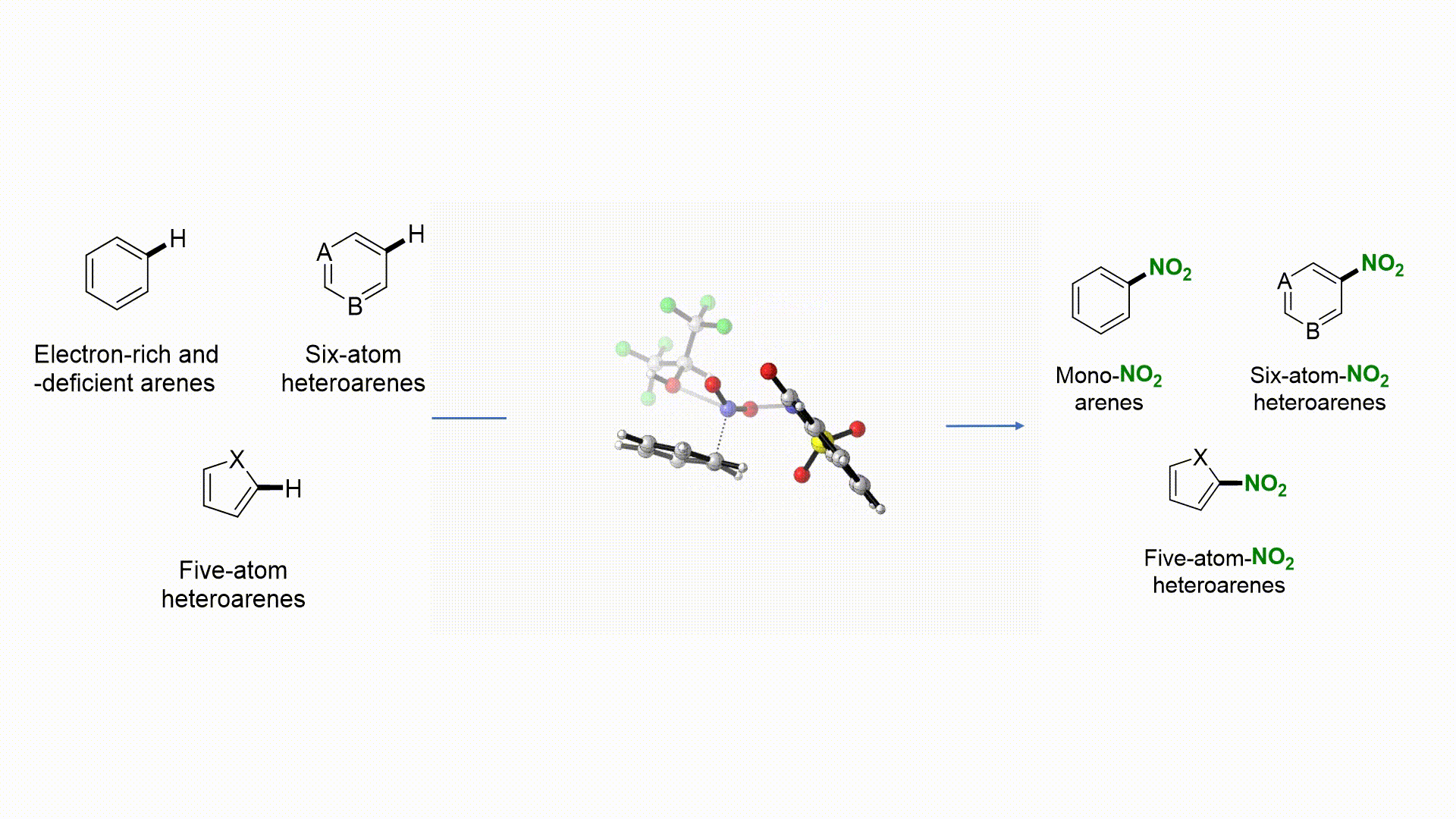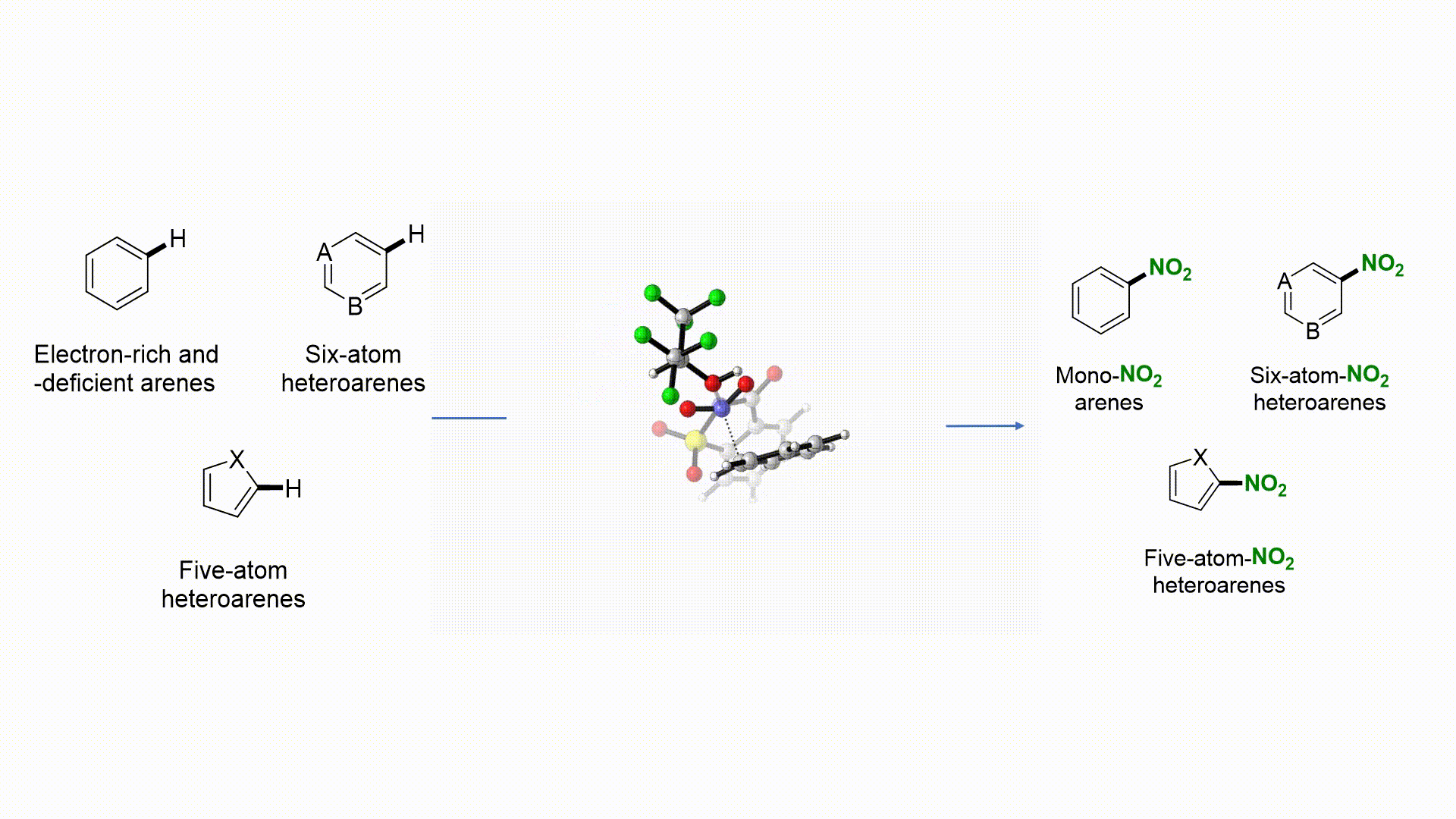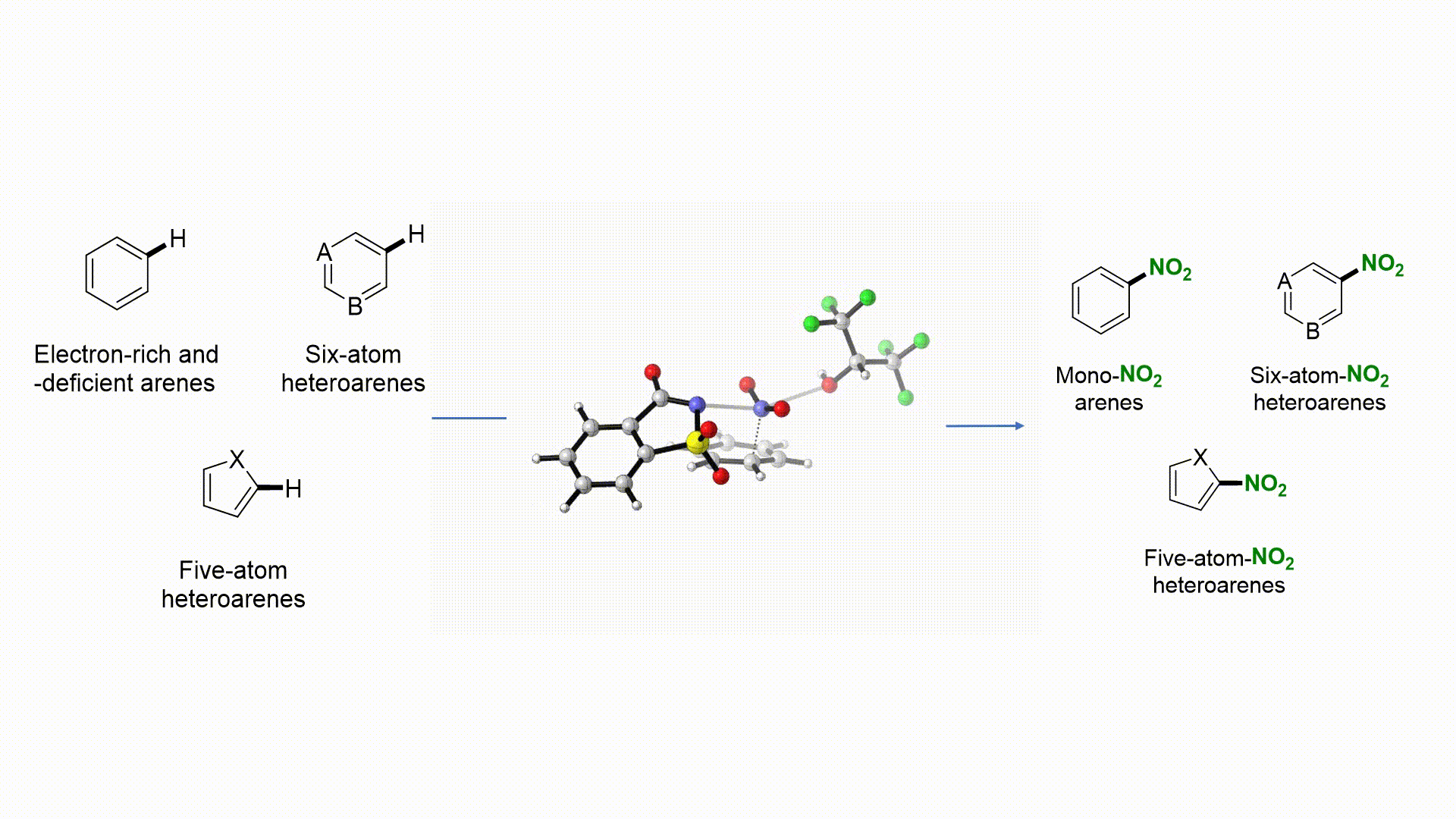
26. Angew. Chem. Int. Ed. 2019, 58, 1447-1450
|
Taming Radical Intermediates for the Construction of Enantioenriched Trifluoromethylated Quaternary Carbon Centers R. Calvo, A. Comas-Vives, A. Togni, D. Katayev German version: Angew. Chem. 2019, 131, 1461-1466 Dedicated to Professor Yuri Belokon on the occasion of his 80th birthday
|

25. ChemPhysChem 2018, 19, 816-821
|
Mapping Perfluoroalkyl Effects in Togni-Type Reagents by Thermolysis N. Santschi, D. Katayev, R. Calvo, B. Jelier
|

24. Wiley Book, Chapter 10, 2018, 409-460
|
Halogenation and Perfluoroalkylation Chemistry Using Hypervalent Halogen Compounds D. Katayev, B. Jelier, A. Togni
|
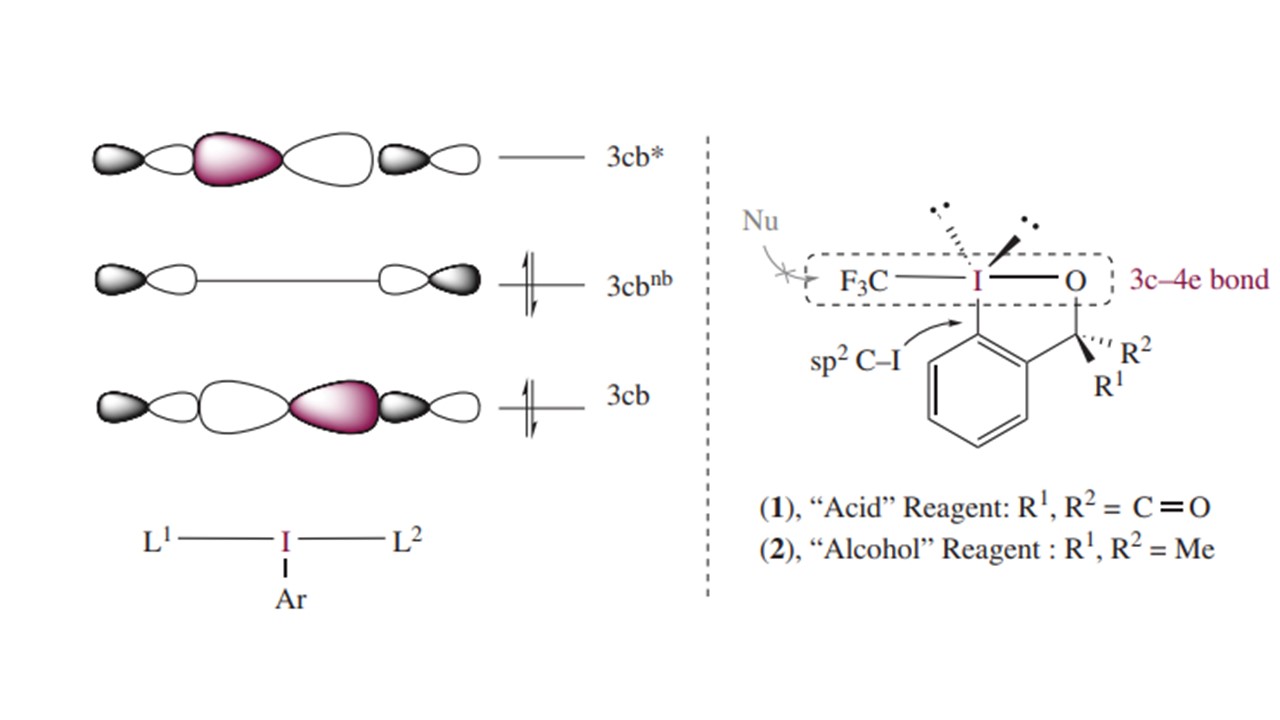
23. Eur. J. Org. Chem. 2018, 31, 4256-4264
|
Facile and Efficient Synthesis of 3‐Pyrimidinyl Oxindoles by Phase‐Transfer‐Catalyzed Regioselective Nucleophilic Aromatic Substitution F. Brüning, D. Katayev, A. Togni
|
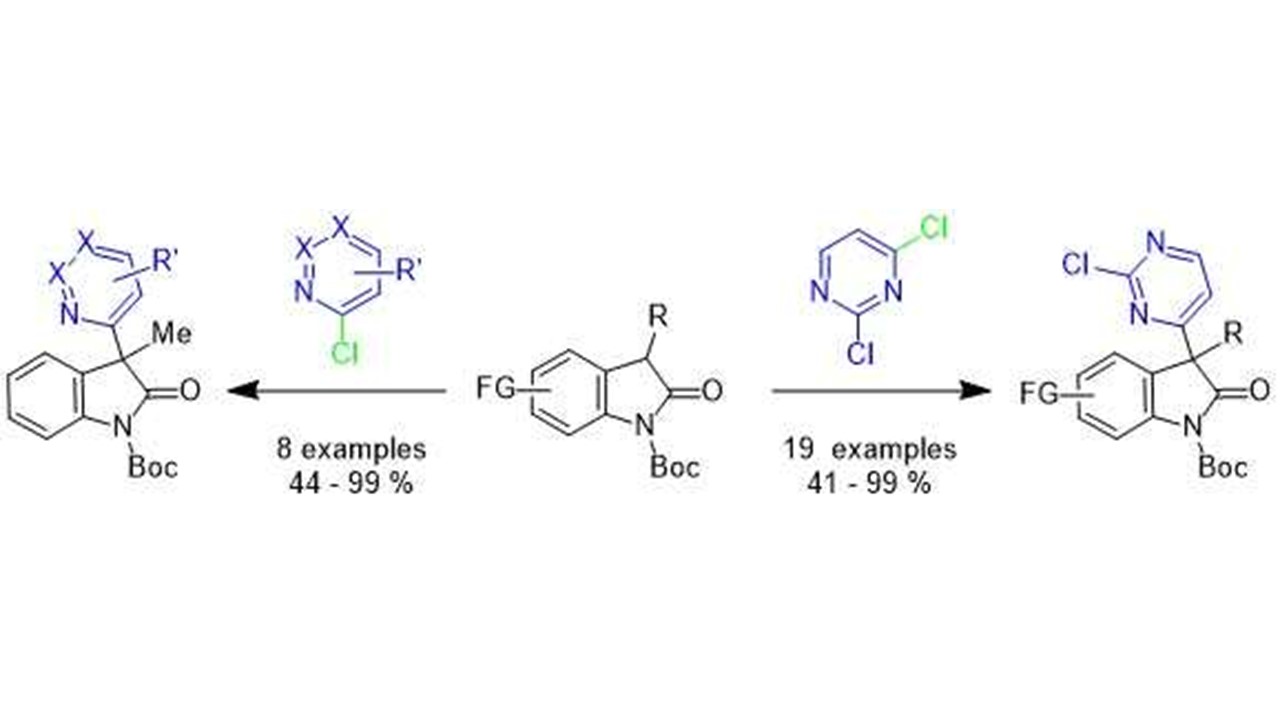
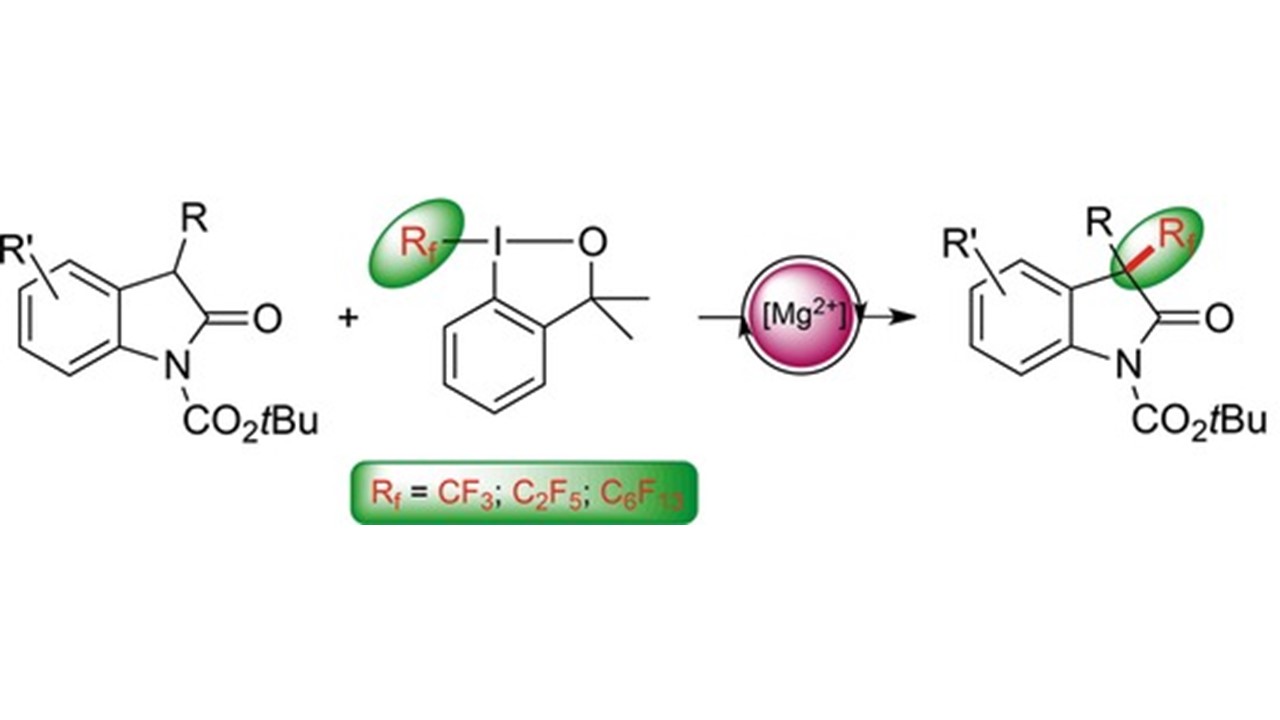
22. Chem. Eur. J. 2017, 23, 8353-8357
|
Magnesium-Catalyzed Electrophilic Trifluoromethylation: Facile Access to All-Carbon Quaternary Centers in Oxindoles D. Katayev, H. Kajita, A. Togni, Highlighted by SYNFACTS, 2017 Author’s profile: Chem. Eur. J. 2017, 23, 8324 Front cover Chem. Eur. J. 2017, 23, 8322 |
PUBLICATIONS FROM POSTDOCTORAL, GRADUATE, AND UNDERGRADUATE STUDIES
21. Chem. Commun. 2016, 52, 4049-4052
|
Synthesis of quaternary α-perfluoroalkyl lactams via electrophilic perfluoroalkylation D. Katayev, J. Václavík, F. Brüning, B. Commare, A. Togni Highlighted in ChemInform, 2016 |
20. The Catalyst Review 2015, 28, 6-11
|
Decarboxylative and Decarbonylative Reactions: An Industrial Perspective D. Katayev, D. Hackenberger, L. Gooßen Published as a Special Feature article with Cover Profile |

19. Org. Lett. 2015, 17, 5898-5909
|
Lewis Acid-Catalyzed Synthesis of α-Trifluoromethyl Esters and Lactones by Electrophilic Trifluoromethylation D. Katavey, V. Matousek, R. Koller, A. Togni Highlighted in ChemInform, 2016 |
18. ChemCatChem 2015, 7, 2028-2032
|
Synthesis of Biaryls by Decarboxylative Hiyama Coupling D. Katayev, B. Exner, L. J. Gooßen Highlighted in ChemInform, 2015 |
17. Chem. Eur. J. 2014, 20, 9902-9905
|
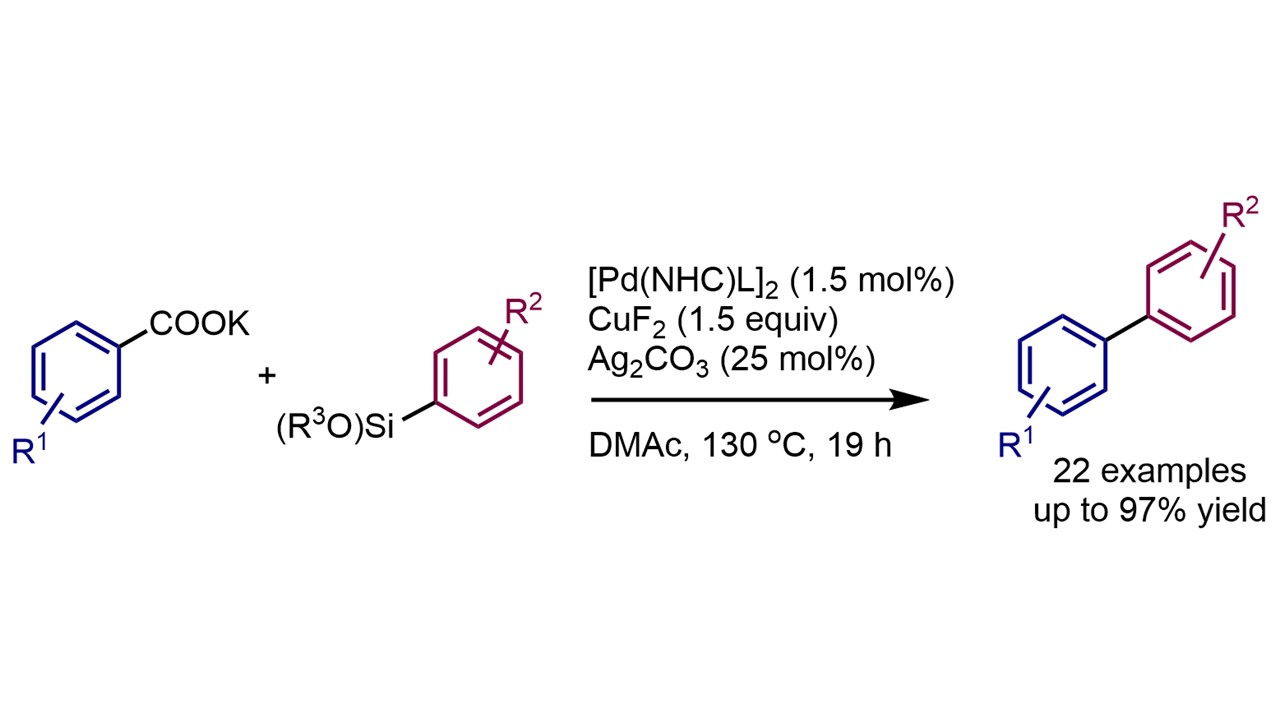
Copper-Mediated ortho-Nitration of (Hetero)Arenecarboxylates D. Katayev, K. F. Pfister, T. Wendling and L. J. Gooßen Highlighted by SYNFACTS, 2014 |

16. Chem. Eur. J. 2014, 20, 15021-15030
|
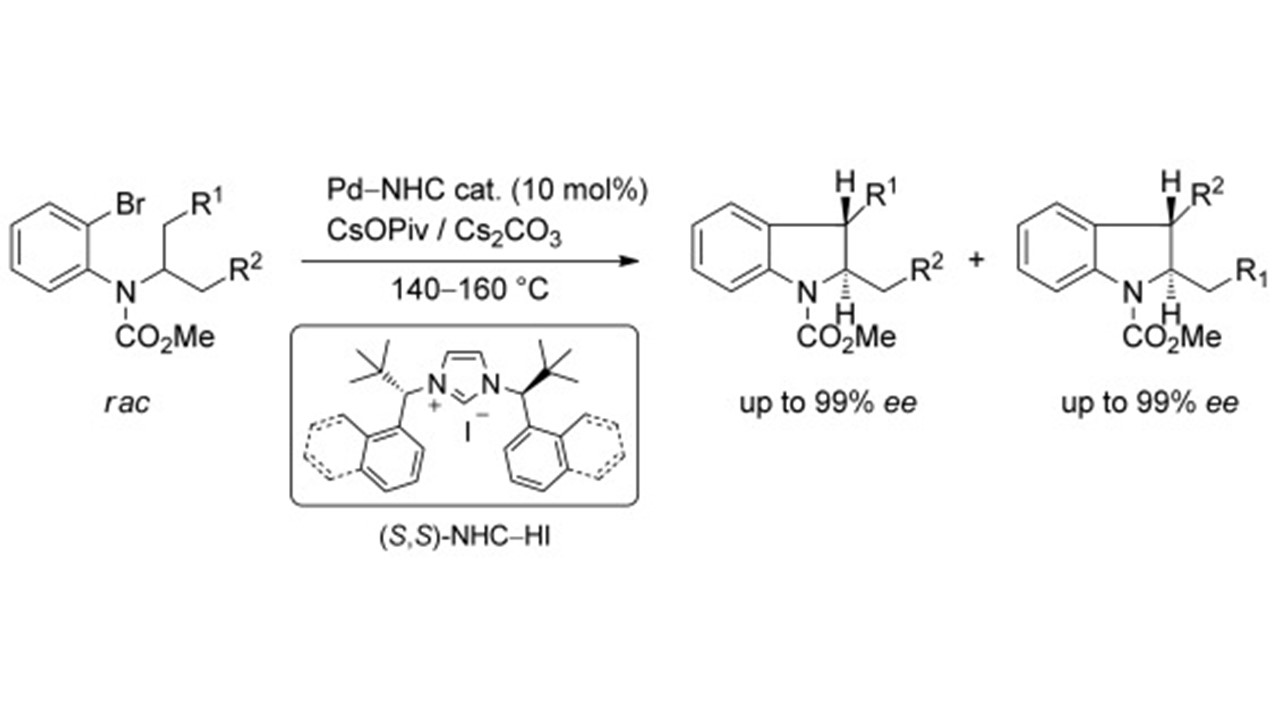
Palladium–N-Heterocyclic Carbene (NHC)-Catalyzed Asymmetric Synthesis of Indolines through Regiodivergent C(sp³)-H Activation: Scope and DFT Study D. Katayev, E. Larionov, M. Nakanishi, C. Besnard, E. P. Kündig Published with Cover Profile Chem. Eur. J. 2014, 20, 14909 |
15. Angew. Chem. Int. Ed. 2013, 52, 9279-9283
|
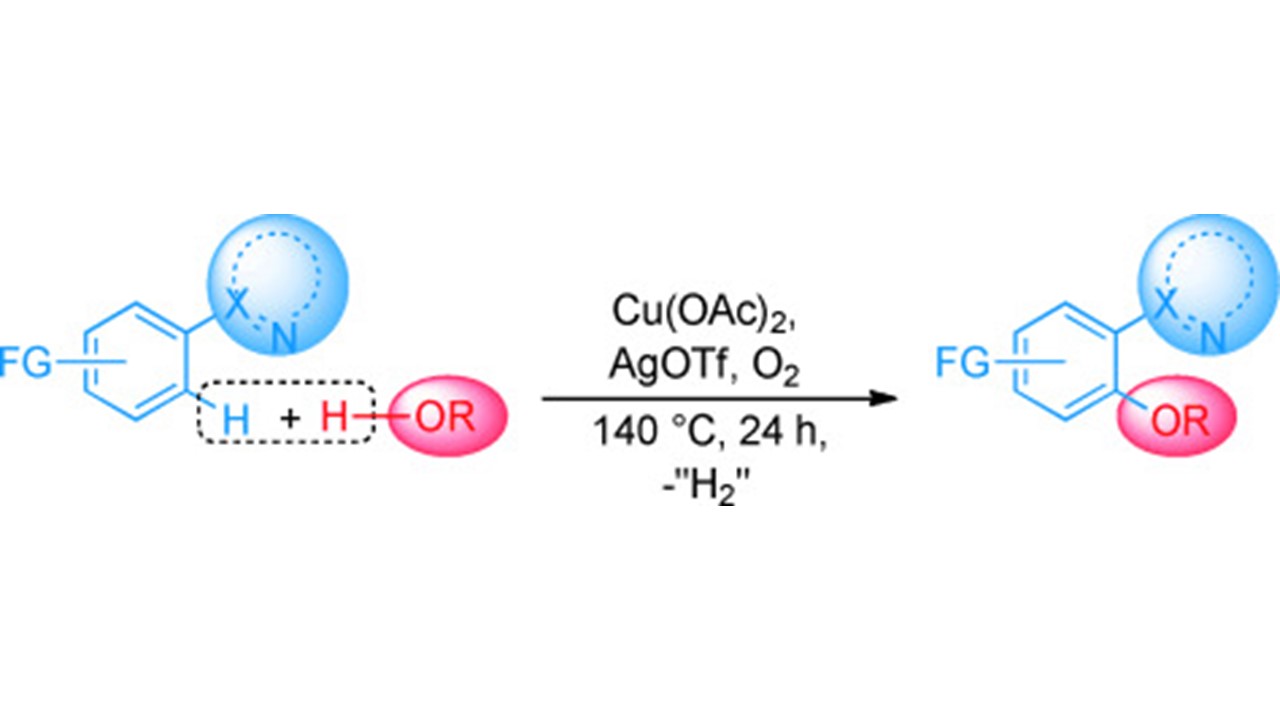
Copper-Catalyzed Dehydrogenative Coupling of Arenes and Alcohols S. Bhadra, C. Matheis, D. Katayev, L. J. Gooßen German version: Angew. Chem. 2013, 125, 9449-9453 |
14. Chem. Eur. J. 2013, 19, 11916-11927
|
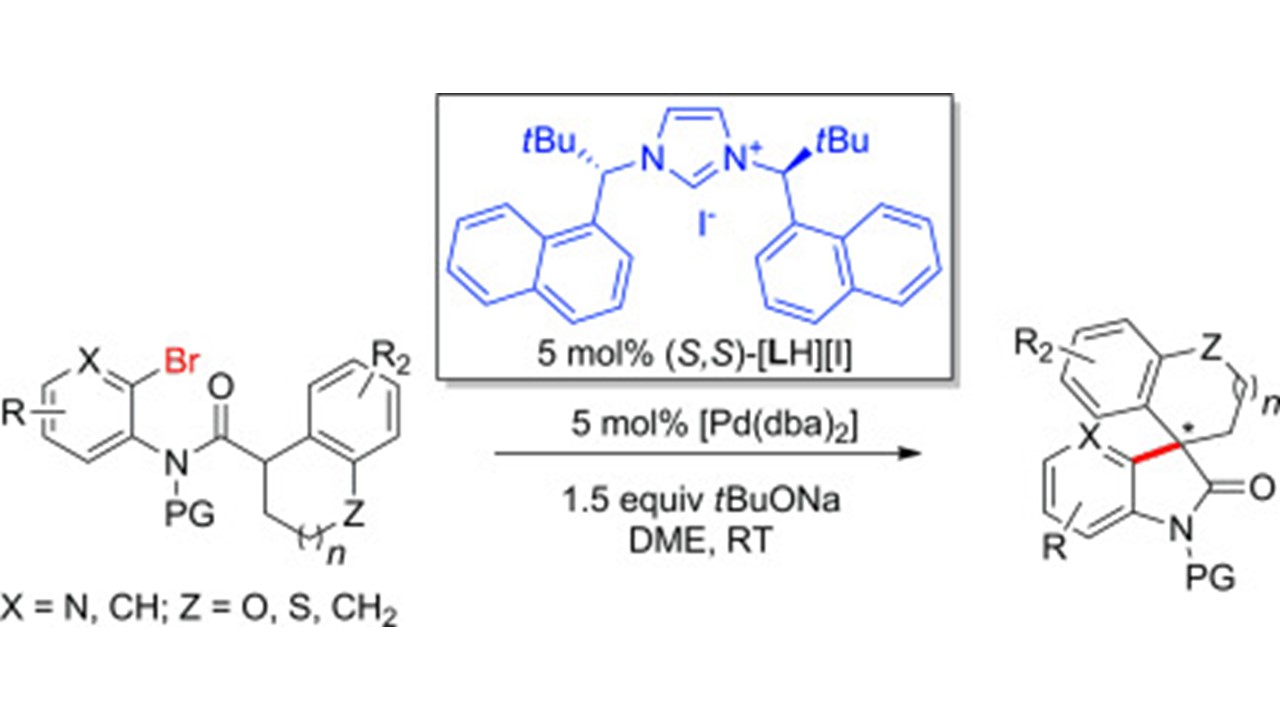
Synthesis of 3,3-Disubstituted Oxindoles by Pd-Catalyzed Asymmetric Intramolecular α-Arylation of Amides. Reaction Development and Mechanistic Studies D. Katayev, D. Banerjee, Y.-X. Jia, C. Besnard, A. K. Sharma, R. B. Sunoj, E. P. Kündig |
13. Chem. Sci. 2013, 4, 1995-2005
|
Scope and Mechanism of Asymmetric C(sp³)-H/C(Ar)-X Coupling Reactions: Computational and Experimental Study E. Larionov, M. Nakanishi, D. Katayev, E. P. Kündig |

12. Helv. Chim. Act. 2012, 95, 2287-2295
|
Catalytic Enantioselective Synthesis of a 3-Aryl-3-benzyloxindole (=3-Aryl-3-benzyl-1,3-dihydro-2H-indol-2-one) Exhibiting Antitumor Activity D. Katayev, E. P. Kündig Special Prof. Dieter Seebach issue |

11. Pure Appl. Chem. 2012, 84, 1741-1748
|
Asymmetric Pd-NHC-Catalyzed Coupling Reactions E. P. Kündig, Y.-X. Jia, D. Katayev, M. Nakanishi |
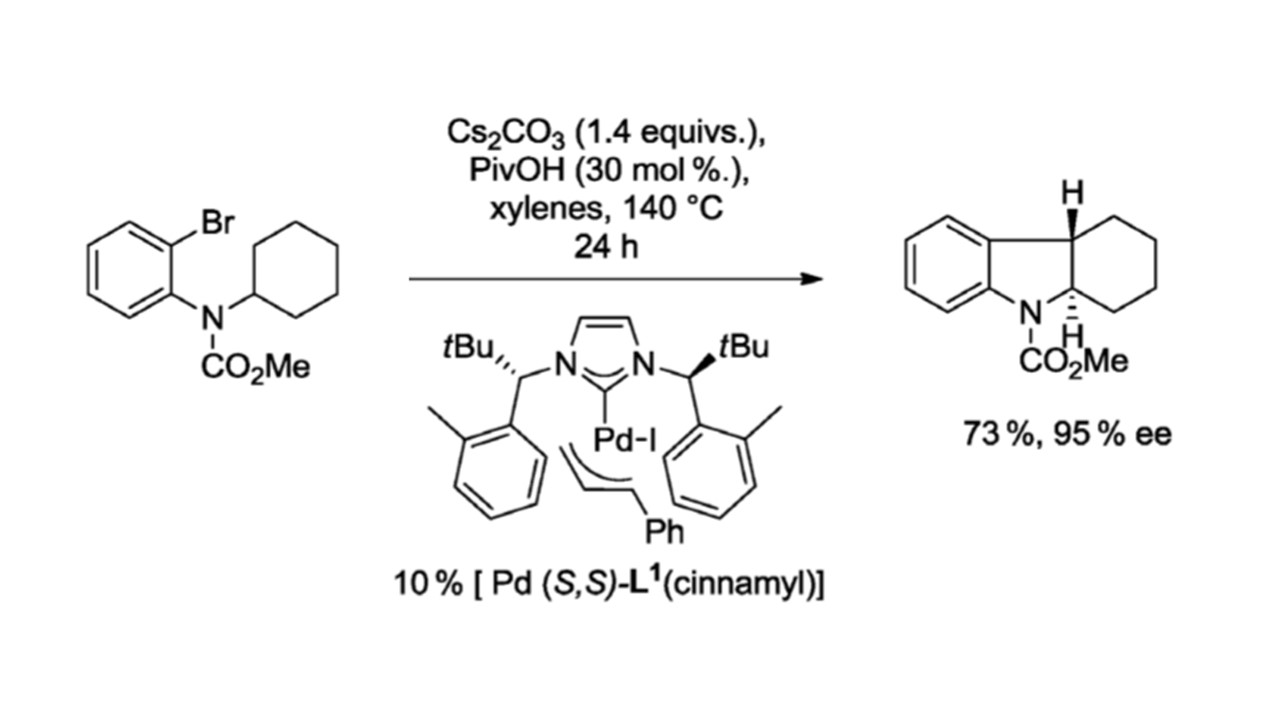
10. Chimia 2012, 66, 241-243
|
Palladium-NHC Catalyzed Enantioselective Synthesis of Fused Indolines via Inert C(sp³)-H Activation M. Nakanishi, D. Katayev, C. Besnard, E. P. Kündig |
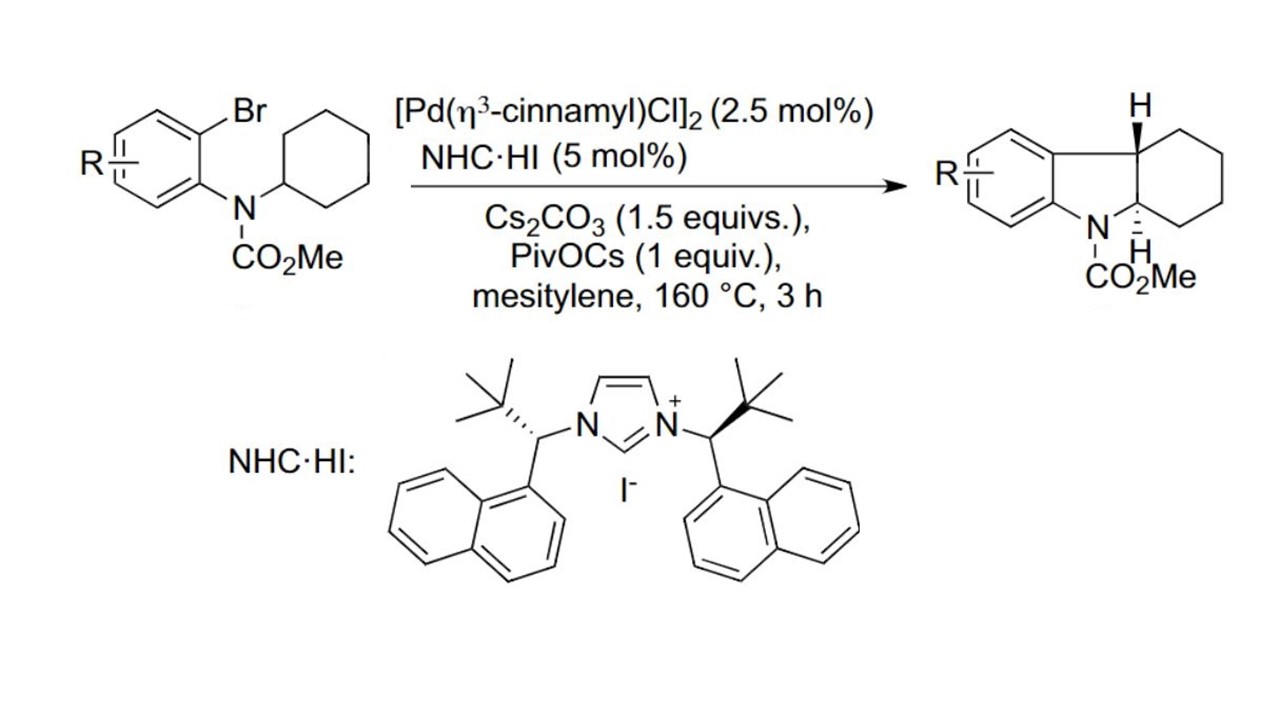
9. Chem. Sci. 2012, 3, 1422-1425
|
Asymmetric C(sp³)-H/C(Ar) Coupling Reactions. Highly Enantioenriched Indolines via Regiodivergent Reactions of a Racemic Mixture D. Katayev, M. Nakanishi, T. Bürgi, E. P. Kündig
|
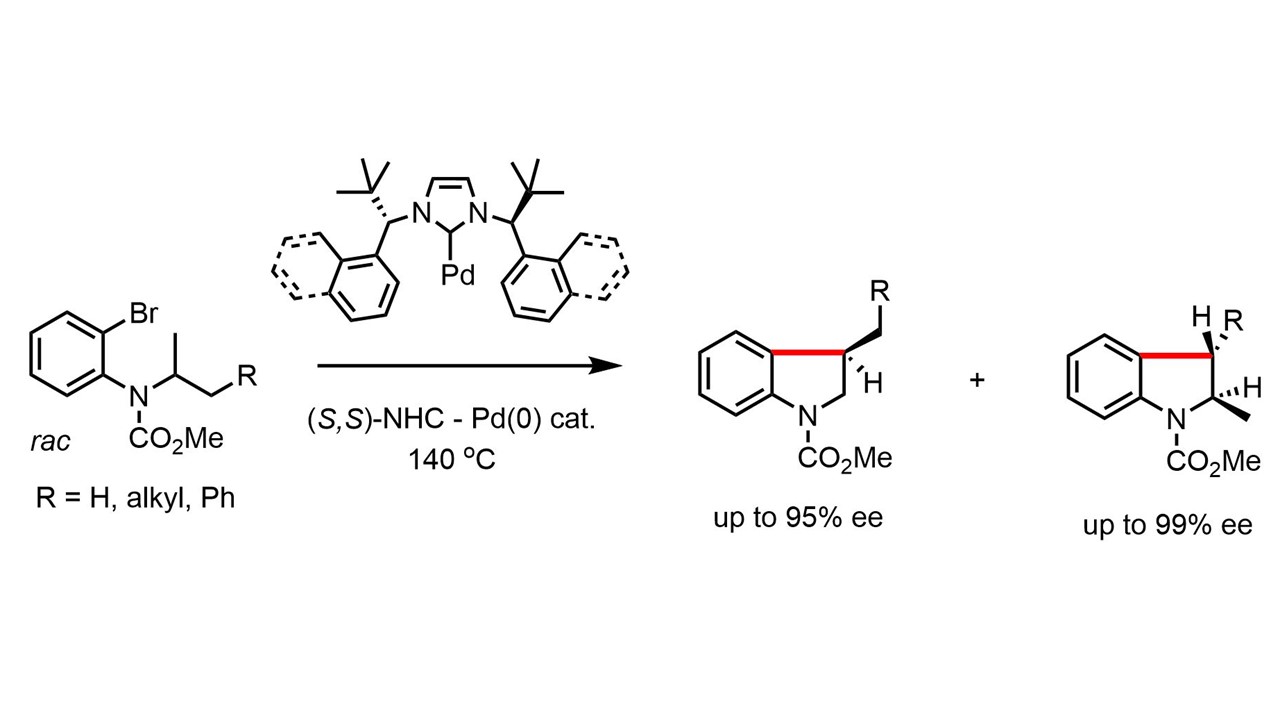
8. Chem. Commun. 2012, 48, 10957-10959
|
Aza-oxindole Synthesis via Base Promoted Truce–Smiles Rearrangement C. Dey, D. Katayev, K. O. Ylijoki, E. P. Kündig
|
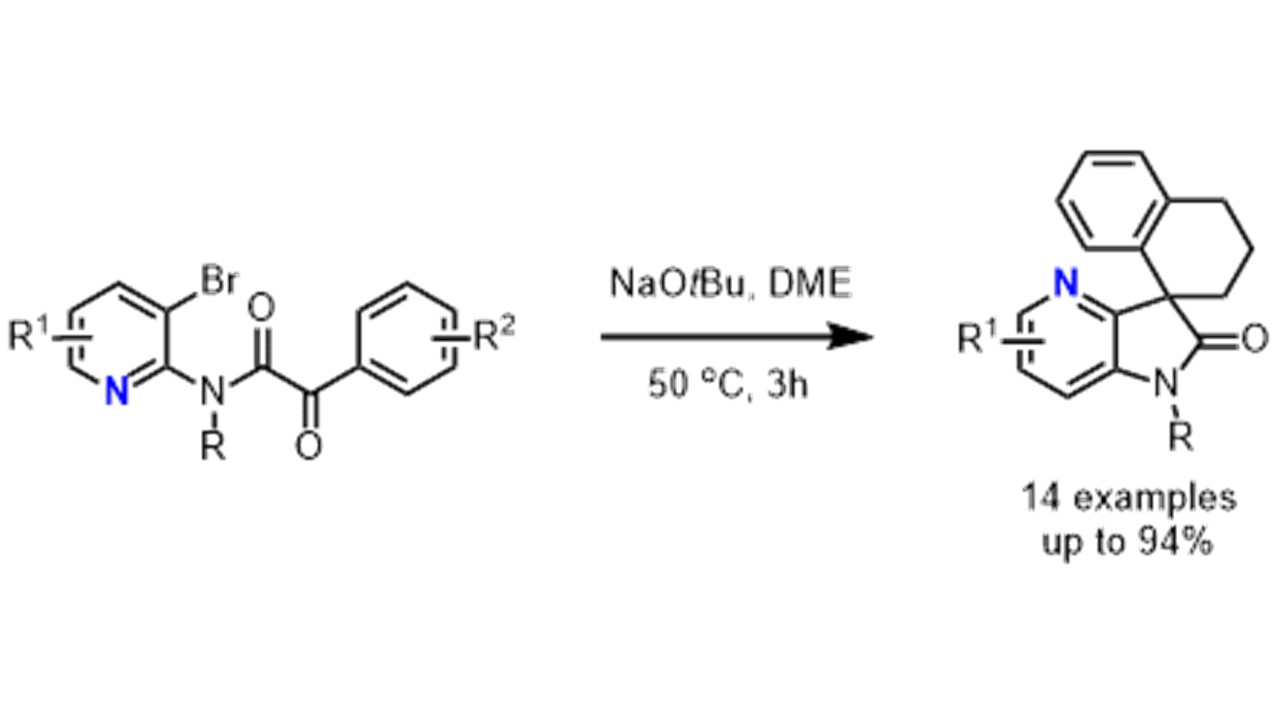
7. Angew. Chem. Int. Ed. 2011, 50, 7438-7441
|
Fused Indolines by Palladium-Catalyzed Asymmetric C-C Coupling Involving an Unactivated Methylene Group M. Nakanishi, D. Katayev, C. Besnard, E. P. Kündig German version: Angew. Chem. 2011, 123, 7576-7579 Published as a Very Important Paper (VIP)
|
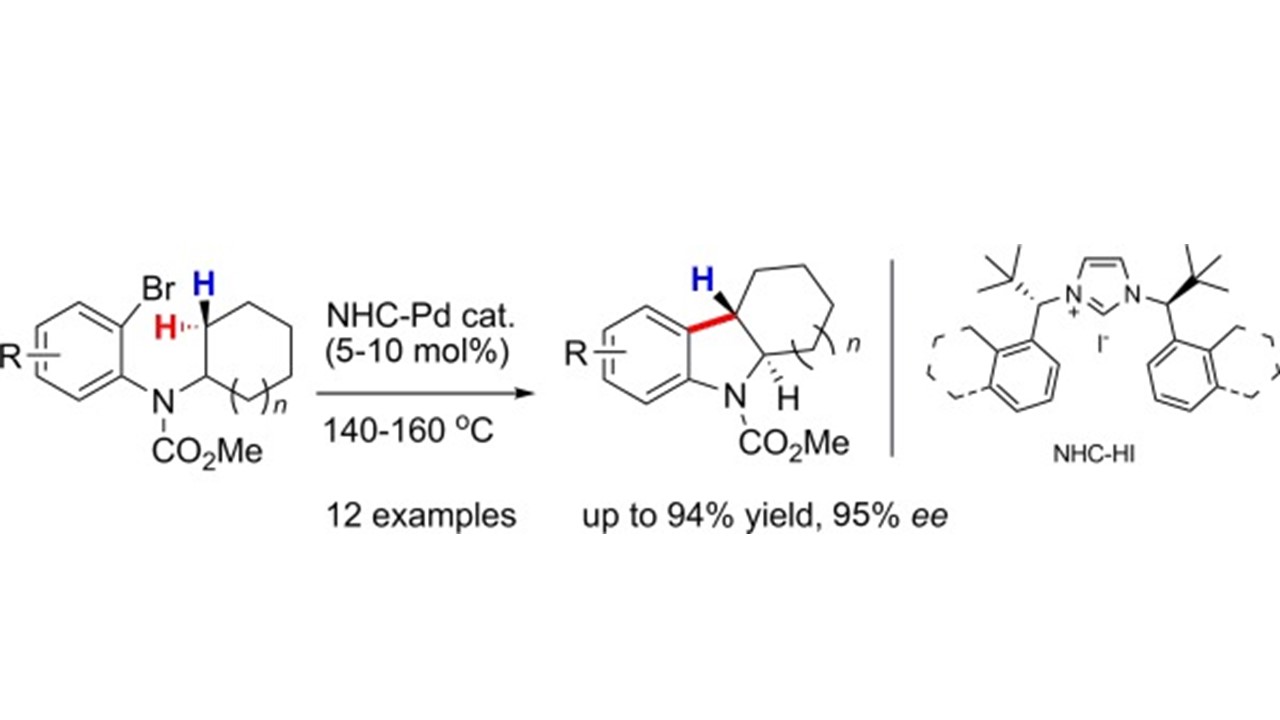
6. Chem. Eur. J. 2010, 16, 6300-6309
|
New Chiral N-Heterocyclic Carbene Ligands in Palladium-Catalyzed α-Arylations of Amides: Conformational Locking through Allylic Strain as a Device for Stereocontrol Y. X. Jia, D. Katayev, G. Bernardinelli, T. Seidel, E. P. Kündig
|

5. Chem. Commun. 2010, 46, 130-132
|
Synthesis of 3-Hydroxyoxindoles by Pd-Catalysed Intramolecular Nucleophilic Addition of Aryl Halides to α- Ketoamides Y. X. Jia, D. Katayev, G. Bernardinelli, T. Seidel, E. P. Kündig
|
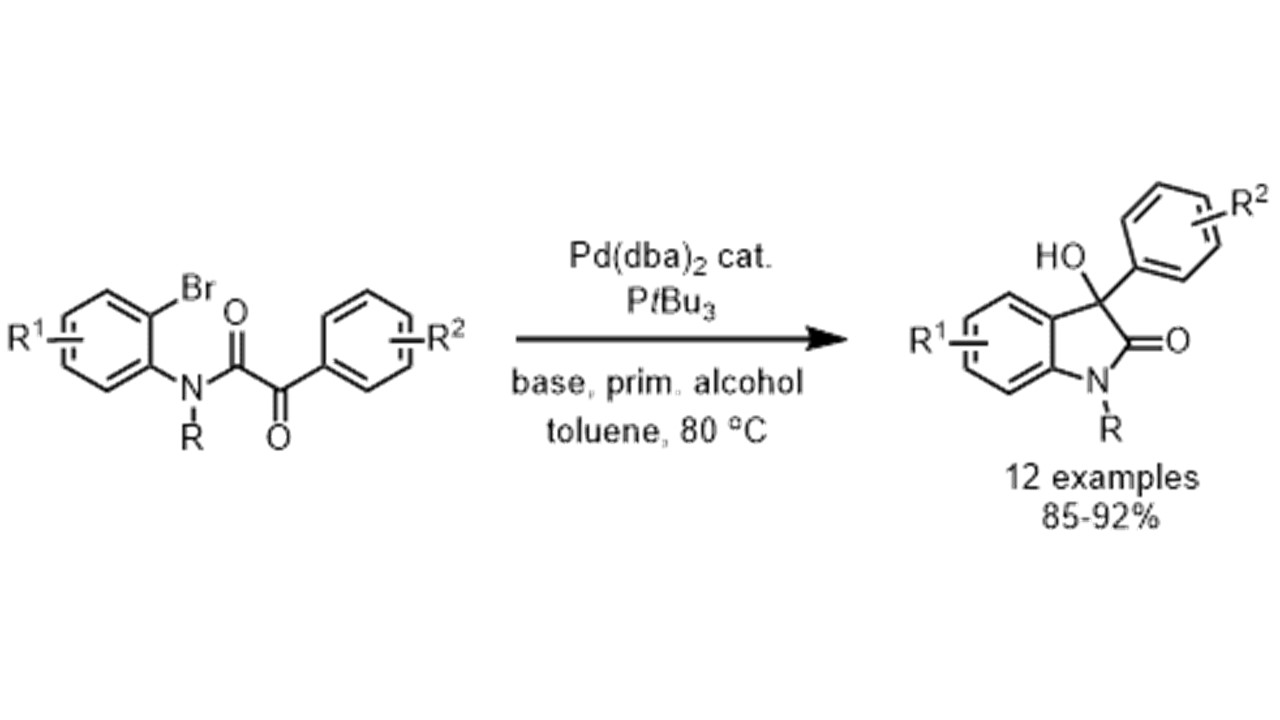
4. Tetrahedron: Asymmetry 2009, 20, 1746-1752
|
Chiral Ion Pairs in Catalysis: Lithium Salts of Chiral Metallocomplex Anions as Catalysts for Asymmetric C-C Bond Formation Y. N. Belokon, V. I. Maleev, D. Katayev, T. F. Saveleva, T. V. Skrupskaya, Y. V. Nelyubina, M. North
|
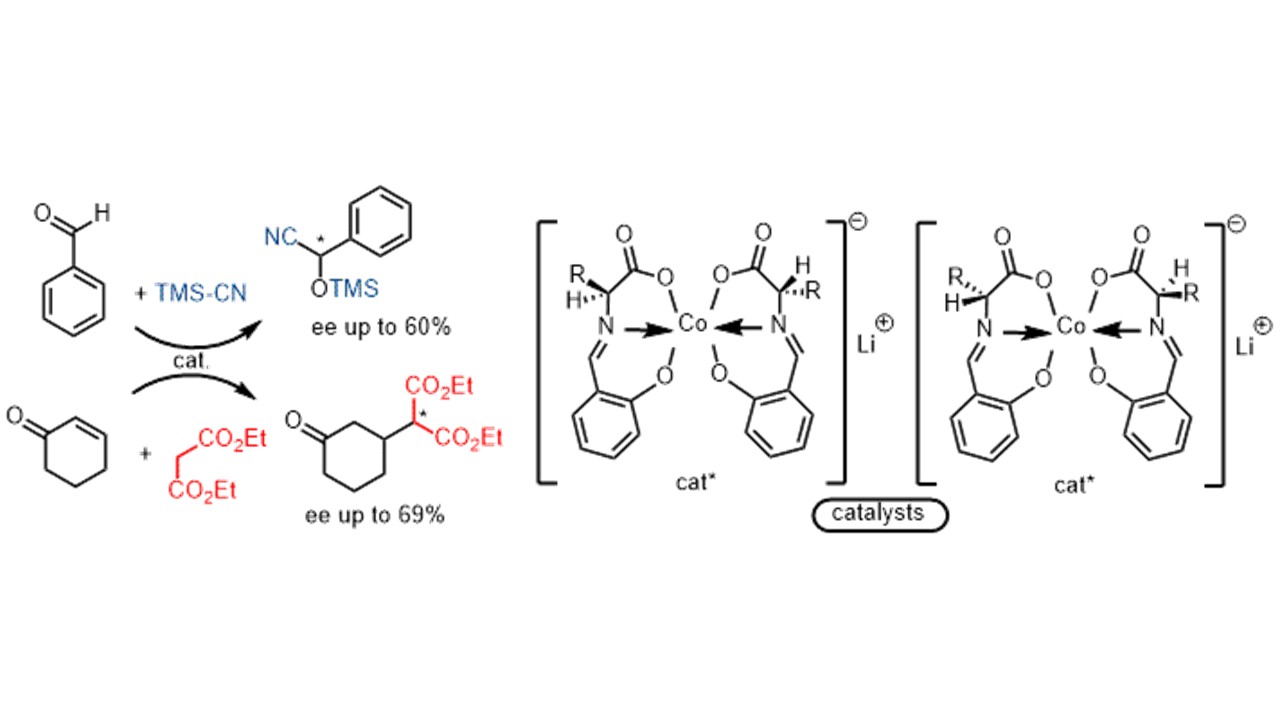
3. Tetrahedron: Asymmetry 2008, 19, 822-831
|
Potassium and Silver Chiral Cobaltate(III) Complexes as Precatalysts for Asymmetric C–C Bond Formation Y.-X. Jia, D. Katayev, G. Bernar Y. N. Belokon, V. I. Maleev, D. Katayev, I. L. Mal’fanov, A. G. Bulychev, T. V. Skrupskaya, K. A. Lyssenko, M. North
|
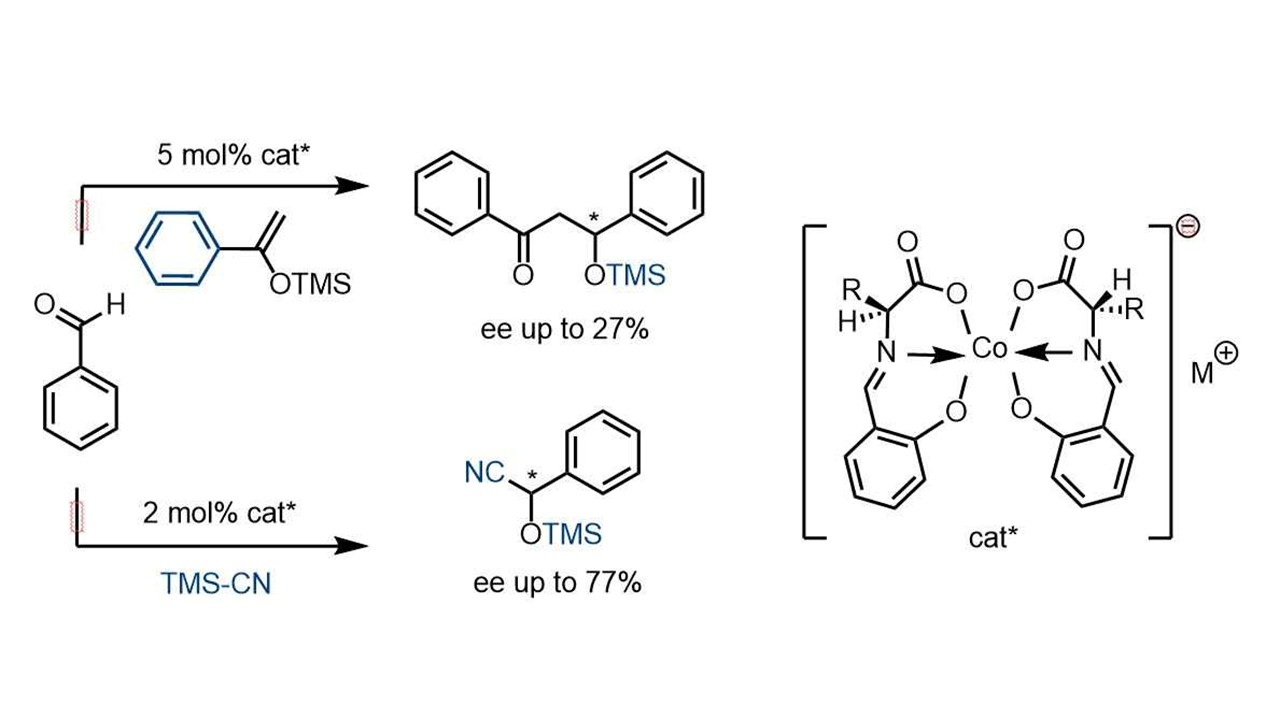
2. Russ. Chem. Bull. 2006, 5, 821-827
|
Anionic Chiral Cobal(III) Complexes as Catalysts of Asymmetric Synthesis of Cyanohydrides Y. N. Belokon, V. I. Maleev, I. L. Malfanov, T. F. Savel’eva, D. Katayev, A. G. Bulychev, D. L. Usanov, M. North
|
1. Tetrahedron: Asymmetry 2006, 17, 2328–2333
|
Chiral Ti(IV) Complexes of Hexadentate Schiff Bases as Precatalysts for the Asymmetric Addition of TMSCN to Aldehydes and the Ring Opening of Cyclohexene Oxide Y. N. Belokon, D. Chusov, D. A. Borkin, L. V. Yashkina, A. V. Dmitriev, D. Katayev, M. North
|